2020 P1 Q16 - Interpreting Mechanism from Energy Profile Diagram
Let Chemistry Guru, Singapore's prestigious JC Chemistry tuition centre, guide you through 2020 A Levels H2 Chemistry Paper 1 Question 16.
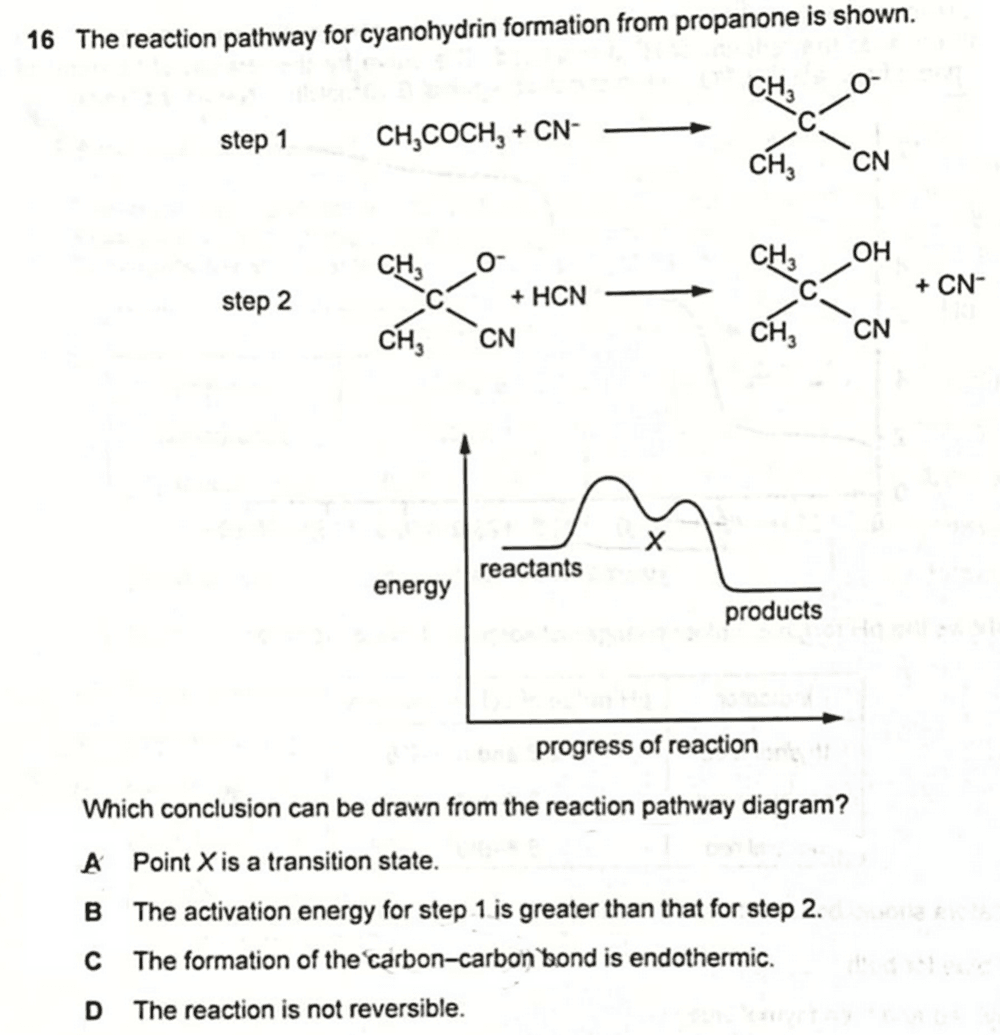
We are asked to draw the correct conclusion about this mechanism from the reaction pathway diagram or energy profile diagram.
Notice the mechanism given is the nucleophilic addition of carbonyl compounds which is in syllabus.
Check out this video to learn how to describe nucleophilic addition mechanism in detail.
Let's run through each of these options and consider their validity.
Option A - Point X is a transition state

A transition state is a theoretical species where the bonds are partially formed and broken as the reactant becomes the product.
It is represented by a maximum point in the energy profile diagram, or the peak at the top of each "mountain".
X is not a transition state. Instead it is an intermediate.
An intermediate is produced during the reaction but is subsequently reacted off in a later step.
Hence option A is not true.
Option B - The activation energy for step 1 is greater than that for step 2
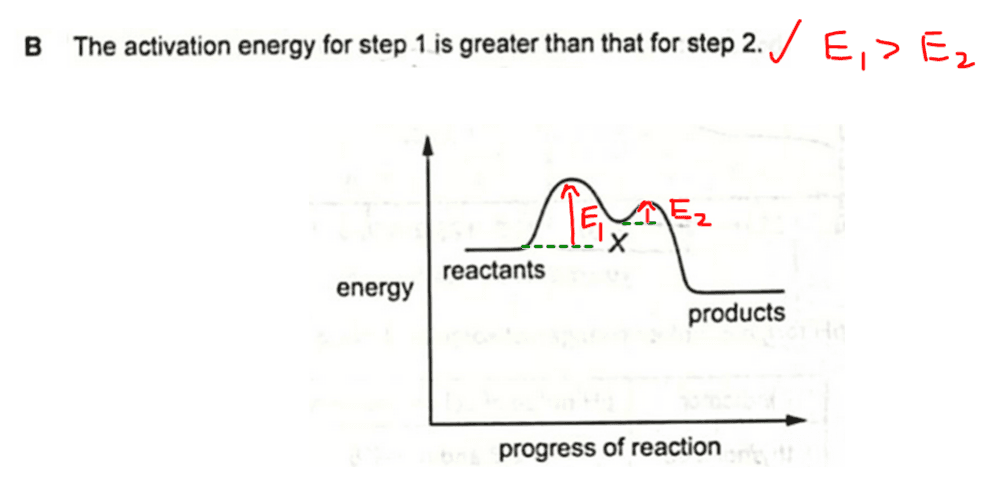
Activation energy is the energy barrier the reactant must possess or overcome in order to proceed to form products.
On the energy profile diagram, activation energy is represented as the energy gap between reactant and transition state.
From the diagram we can see that activation energy for step 1, E1, is greater than activation energy for step 2, E2.
Hence option B is true and is our best answer.
Option C - The formation of the carbon-carbon bond is endothermic

Energy is released when bonds are formed, therefore formation of carbon-carbon bond should be exothermic instead.
Hence option C is not true.
Option D - The reaction is not reversible

The energy profile diagram for irreversible and reversible reactions look the same.
Hence we cannot determine if the reaction is irreversible just by considering the reaction pathway diagram.

Therefore the answer to this question is option B.
Topic: Kinetics, Physical Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2020 A Level H2 Chemistry Paper 1.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's trusted JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
