2020 P1 Q18 - Deduce Number of Chiral Carbons
Let Chemistry Guru, Singapore's prestigious JC Chemistry tuition centre, guide you through 2020 A Levels H2 Chemistry Paper 1 Question 18.
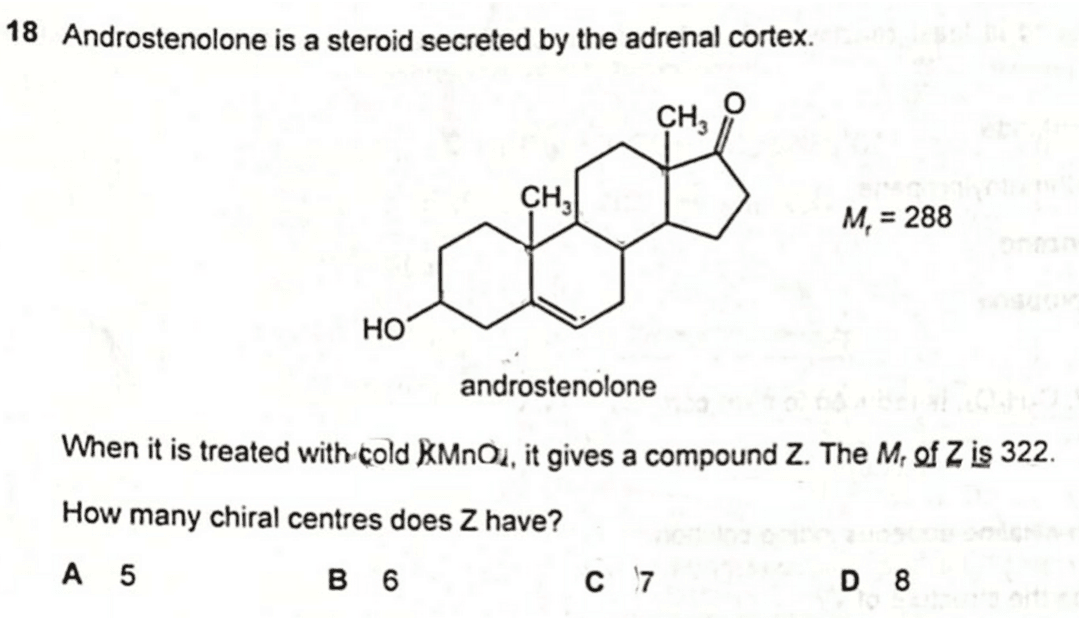
We are required to determine the number of chiral centres for compound Z.
First we have to determine which functional group in reactant androstenolone reacts with cold KMnO4.
There are 2 functional groups that can be oxidised: alkene and secondary alcohol.
Secondary alcohol will only be oxidised when KMnO4 is heated.
Find out more about other reactions of alcohols.
In this case only alkene will react with cold KMnO4 to form diol.
Check out all the reactions of alkenes.
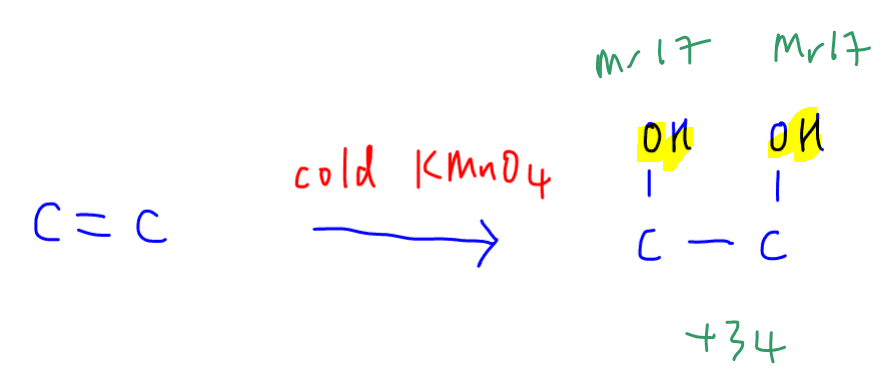
Notice there is an increase in the molar mass by 34 due to addition of 2 OH groups.
We can now deduce the structure of product Z.
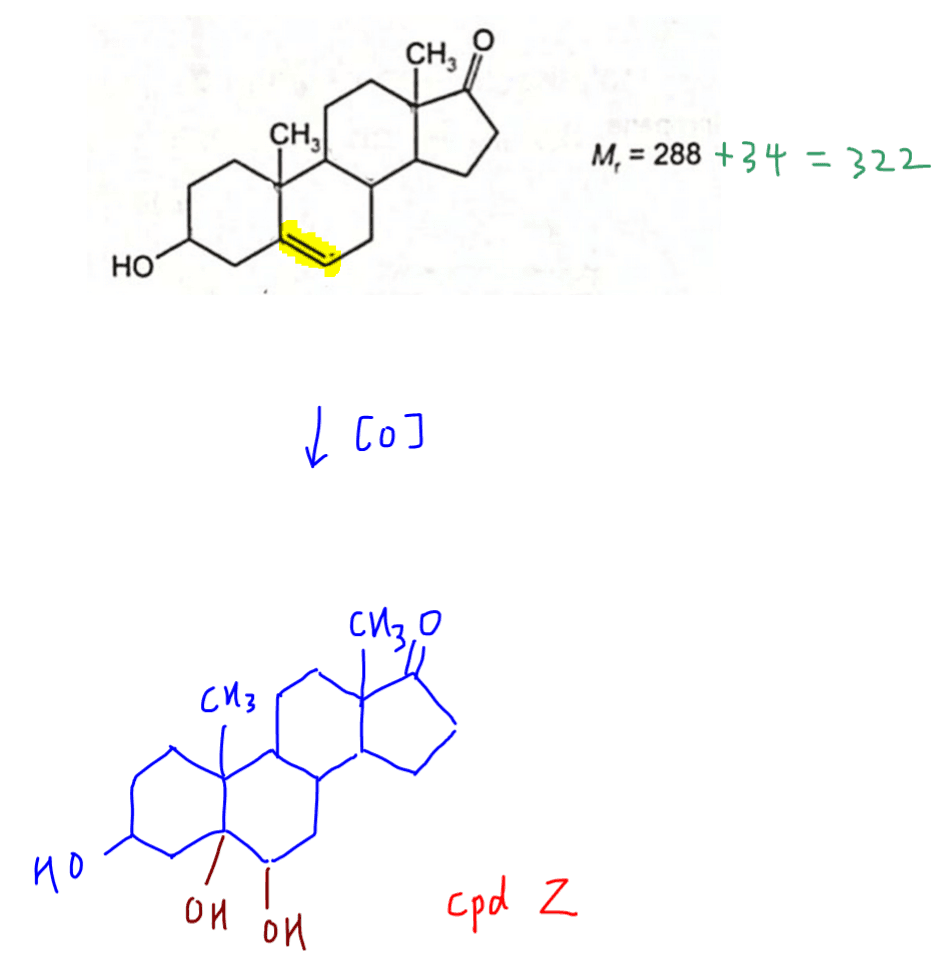
If we compare molar mass of reactant androstenolone and product Z, there is an increase in molar mass by 34.
This is consistent with our deduction that only alkene is oxidised.
We can now determine the number of chiral carbons in Z.
A chiral carbon is saturated and bonded to 4 different groups.
Therefore it is asymmetrical.
We can identify chiral carbons quickly in a skeletal formula by focusing on carbons bonded to 3 groups (with a hidden hydrogen) and 4 groups.
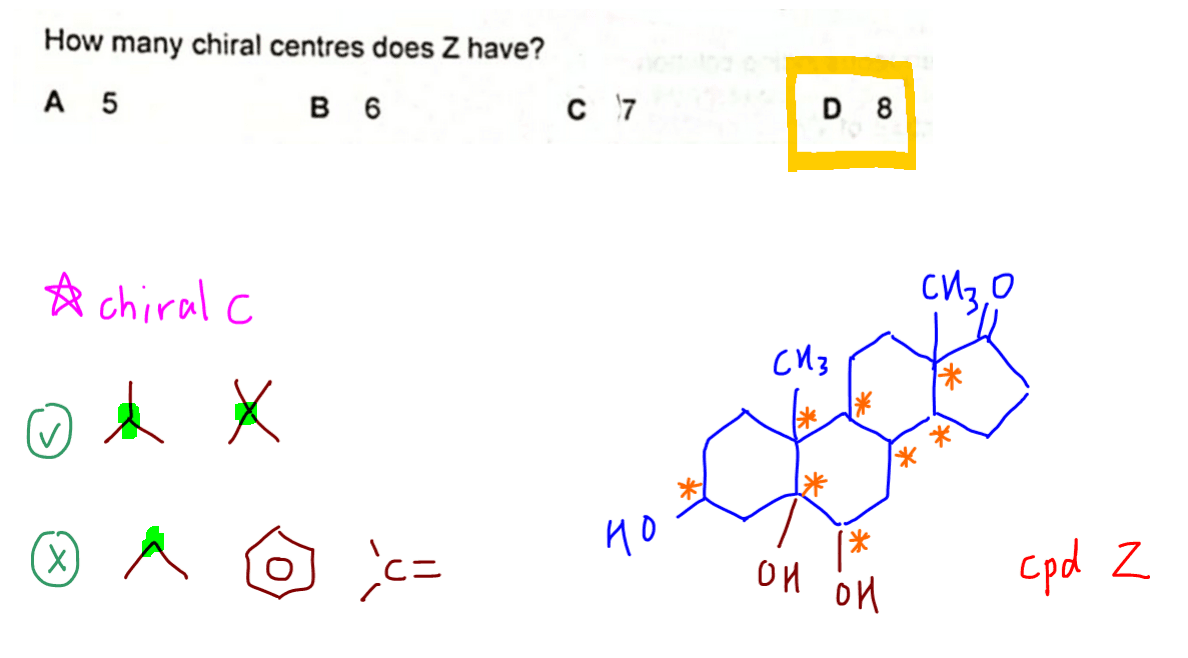
In this case we can find 8 chiral carbons in Z and the answer to this question is option D.
Topic: Intro to Organic Chem & Alkenes, Organic Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2020 A Level H2 Chemistry Paper 1.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's trusted JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
