2020 P1 Q20 - Deducing Reactions in Catalytic Converter
Let Chemistry Guru, Singapore's prestigious JC Chemistry tuition centre, guide you through 2020 A Levels H2 Chemistry Paper 1 Question 20.

We are required to deduce the reactions occurring in the catalytic converter.
Here is what would happen when pollutants from exhaust fumes pass through the catalytic converter.
In general the pollutant gases are converted to gases with no adverse effect on the environment.
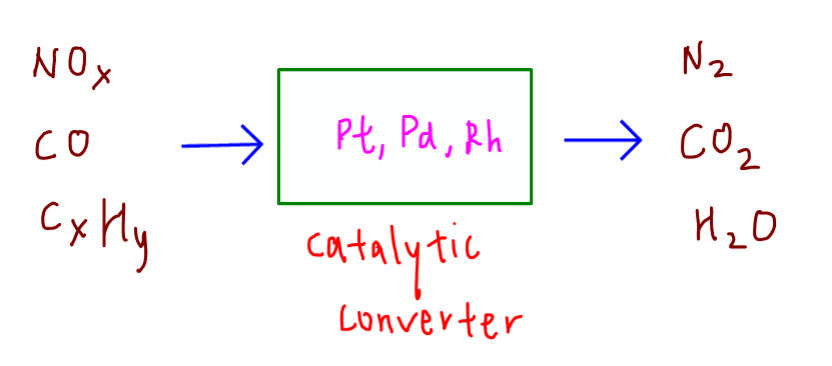
Oxides of nitrogen are converted to nitrogen gas N2.
Carbon monoxide and unburnt carbon are converted to carbon dioxide CO2.
CO2 is a major contributor to global warming but only when in excessive quantities.
Compared to other carbon-containing pollutant gases, CO2 is generally harmless and very stable.
Unburnt hydrocarbons are converted to carbon dioxide and water.
Let's run through the statements and see if they are correct.
1. Carbon dioxide is removed by reduction.
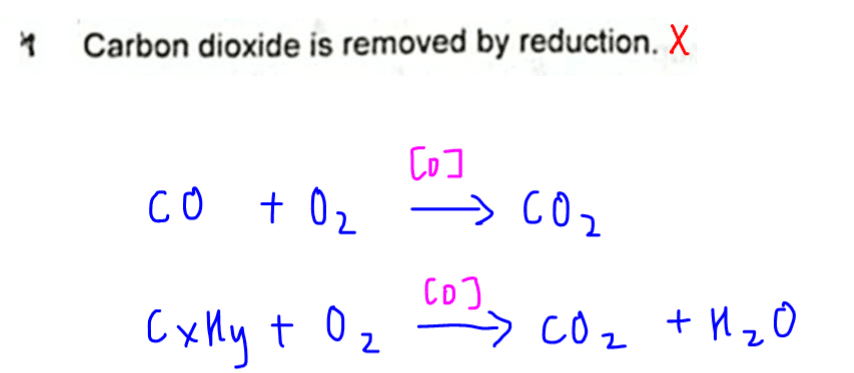
This is not true as the catalytic converter converts more harmful compounds like carbon monoxide, unburnt carbon and hydrocarbons to carbon dioxide.
CO2 is produced instead of being removed.
2. Oxides of nitrogen are removed by reduction.
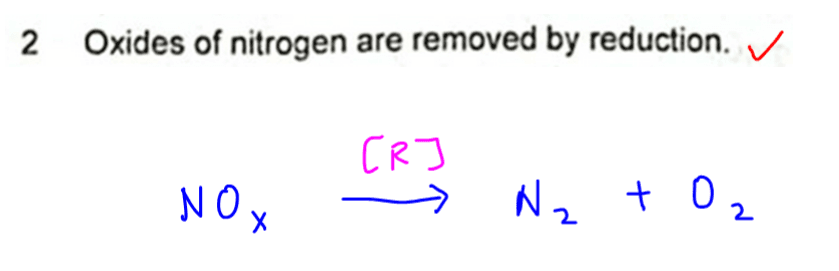
Oxides of nitrogen are converted to N2 gas.
Since oxygen is removed, this is considered as reduction.
Hence statement 2 is correct.
3. Unburnt hydrocarbons are removed by oxidation.
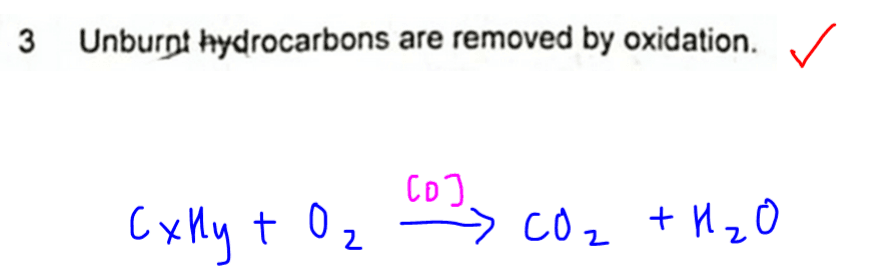
This is true as conversion of hydrocarbons to CO2 and H2O is considered as combustion.
Hence hydrocarbons are oxidised.
We can now run through the options and determine the answer to this question is option B.

Topic: Kinetics, Physical Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2020 A Level H2 Chemistry Paper 1.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's trusted JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
