2020 P1 Q25 - Deducing Name and Reactions of Amide
Let Chemistry Guru, Singapore's top choice for JC Chemistry tuition, guide you through 2020 A Levels H2 Chemistry Paper 1 Question 25.

We are asked to determine which of the following statements about amide C3H7CONH2 is correct.
A. Its name is propanamide.

There are 4 carbons in this amide so its name should be butanamide instead.
Hence statement A is not true.
B. When heated with NaOH(aq), it will form sodium propanoate.

Since we know the amide is butanamide, it will form sodium butanoate when hydrolysed in alkaline medium.
Hence statement B is not true.
C. When heated with H2SO4(aq), it will form butanoic acid.
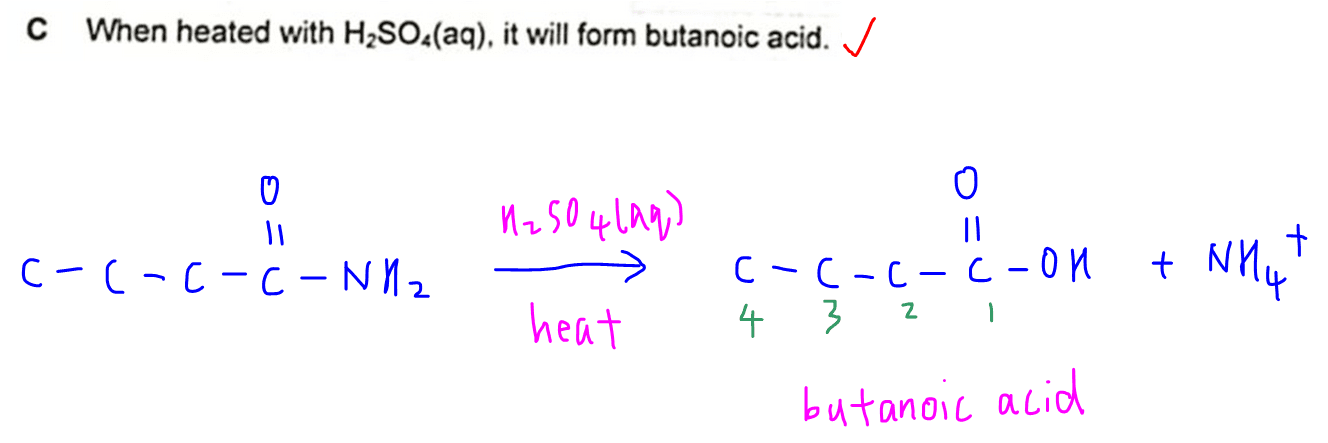
On acidic hydrolysis, butanamide will form butanoic acid and ammonium ion.
Hence statement C is true.
D. It can be formed using butanoic acid and NH3(aq) at room temperature.
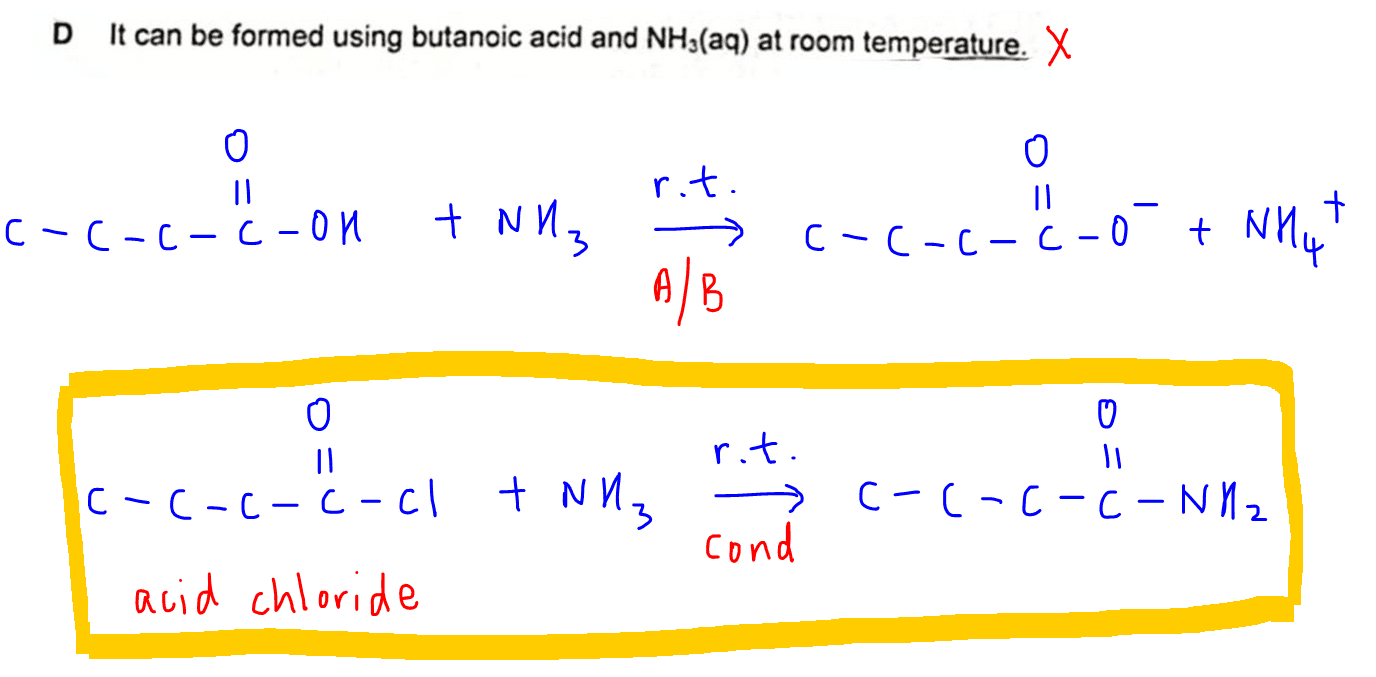
When NH3(aq) is added to butanoic acid at room temperature, neutralisation will take place instead of condensation.
This is because neutralisation occurs at much milder conditions.
The products will be ammonium butanoate instead of butanamide.
In order to form butanamide, we need to convert butanoic acid to butanoyl chloride using PCl5 or SOCl2 anhydrous.
Then conc NH3 can be added to butanoyl chloride at room temperature to form butanamide.
Hence statement D is not true.
We can now look at our options and determine that option C is the best answer.

Here are the must-know reactions of nitrogen compounds such as amine, phenylamine and amides for A Level Chemistry syllabus.
Topic: Nitrogen Compounds, Organic Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2020 A Level H2 Chemistry Paper 1.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's esteemed H2 Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
