2020 P1 Q27 - Hydrogen Oxygen Fuel Cell in Alkaline Medium
Let Chemistry Guru, Singapore's top choice for JC Chemistry tuition, guide you through 2020 A Levels H2 Chemistry Paper 1 Question 27.
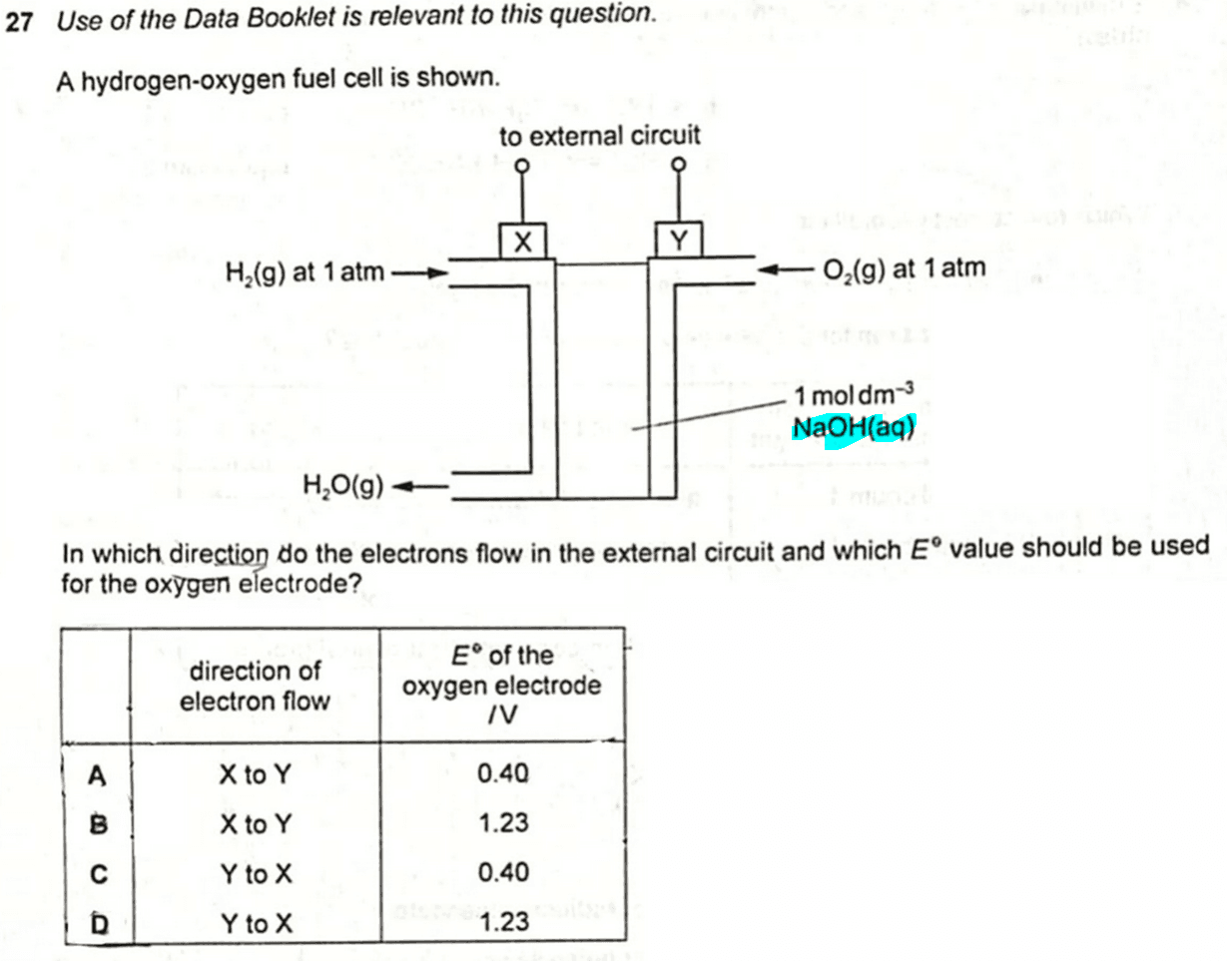
We are given a hydrogen-oxygen fuel cell with NaOH as electrolyte.
We need to determine the direction of electron flow in the external circuit and the E value for the oxygen electrode.
Let us first deduce the anode and cathode from the redox reaction between hydrogen and oxygen.
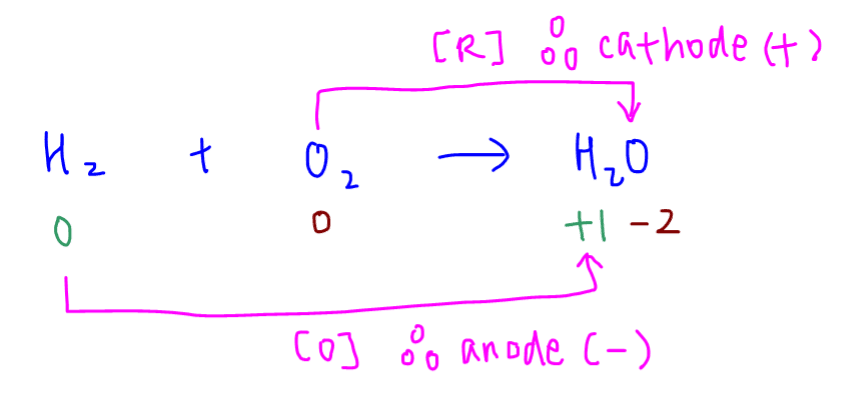
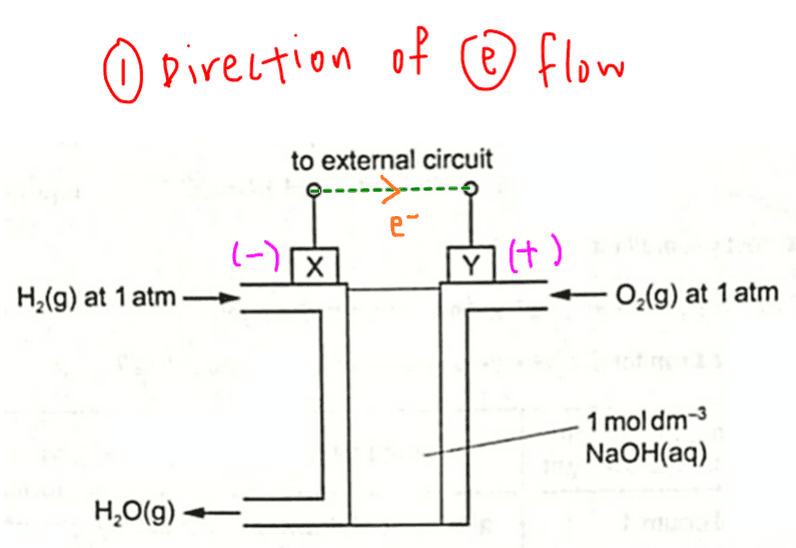
Hydrogen is oxidised to water so electrode X at the hydrogen side is the anode (negatively charged).
Oxygen is reduced to water so electrode Y at the oxygen side is the cathode (positively charged).
So in the external circuit, electron will flow from the negative electrode X to positive electrode Y.
Next let us determine the correct half equation and E value for the oxygen electrode.
Since oyxgen is reduced, it should be found on the left hand side of the half equation found in the data booklet.
The electrolyte is NaOH(aq), so the reaction is in alkaline medium.
Hence we want the half equation where O2 is reduced to a product with stable oxidation state, in the presence of OH-.
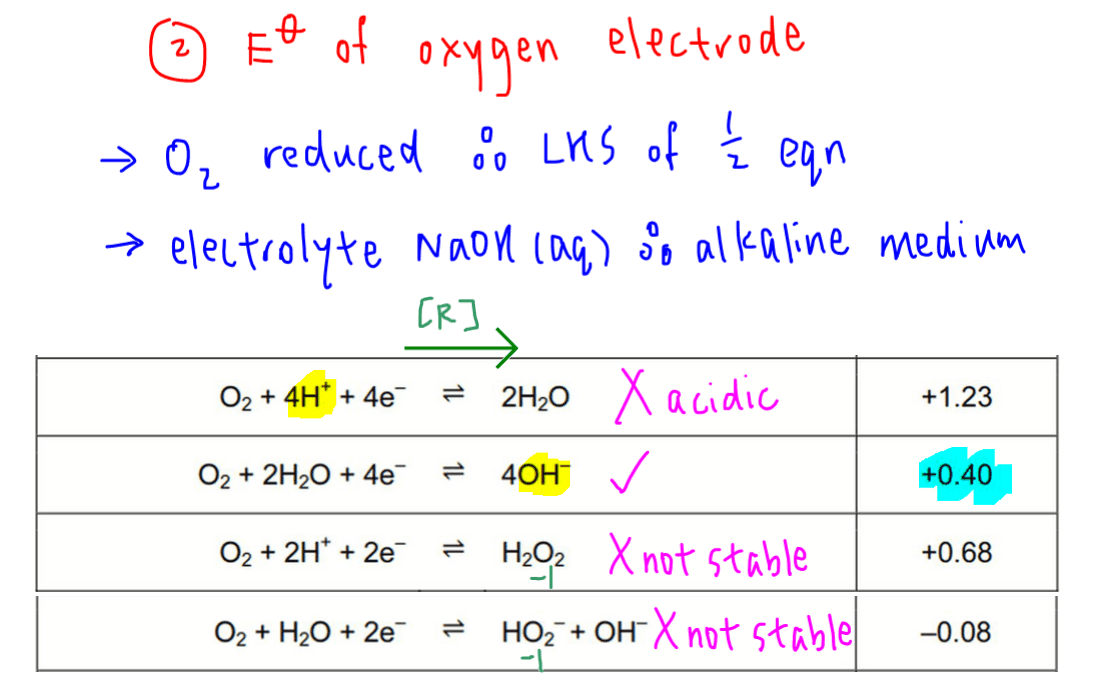
Comparing the options, the following half equation is the best choice since OH- is present and oxidation state of oxygen in product OH- is -2 and stable.
O2 + 2H2O + 4e ↔ 4OH- +0.40V
Finally we can compare the options and determine the answer to this question is option A.

Topic: Electrochemistry, Physical Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2020 A Level H2 Chemistry Paper 1.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's esteemed H2 Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
