2020 P1 Q5 - Deducing Temperature and Boiling Point of Mixture
Let Chemistry Guru, Singapore's renowned JC Chemistry tuition centre, guide you through 2020 A Levels H2 Chemistry Paper 1 Question 5.

We are required to determine the initial temperature of the mixture and its boiling point.
Let's consider the process of mixing 2 liquids.
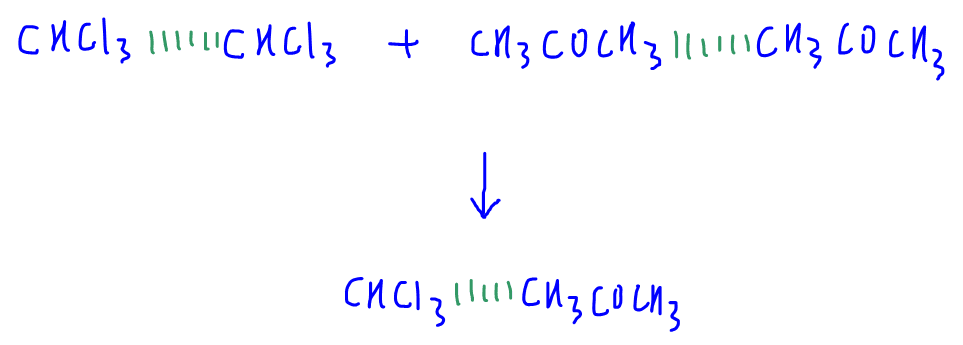
CHCl3 and CHCl3 molecules are separated, while CH3COCH3 and CH3COCH3 molecules are separated.
Hence energy is required to overcome the attraction between these molecules.
On mixing CHCl3 and CH3COCH3 molecules, energy is released from the interactions formed between them.
It is stated that stronger intermolecular forces are formed between CHCl3 and CH3COCH3 molecules.
Therefore more energy is released than energy required and the process is exothermic.
This means heat is released during mixing which increases the temperature of the mixture to above 20oC.
Also, more energy is required to overcome the stronger attraction between the mixture, hence boiling point will be higher than that of CHCl3 at 61oC.

We can now look at the options and figure out the answer.
Initial temperature of mixture is above 20oC while the boiling point of mixture is above 61oC.

Hence the answer to this question will be option A.
Topic: Intermolecular Forces, Physical Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2020 A Level H2 Chemistry Paper 1.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's trusted JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
