2020 P1 Q9 - Determine Empirical Formula of Boron Carbide
Let Chemistry Guru, Singapore's renowned JC Chemistry tuition centre, guide you through 2020 A Levels H2 Chemistry Paper 1 Question 9.

We are required to determine the empirical formula of boron carbide given the mass analysis of its combustion to form boron oxide and carbon dioxide.
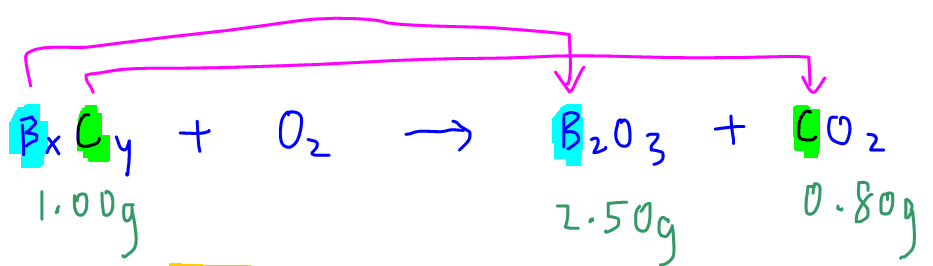
Notice all the boron in boron carbide is transferred to B2O3 hence the amount of boron in 1.00g of boron carbide is equal to the amount of boron in 2.50g of B2O3.
Similarly all the carbon is transferred to CO2 and the amount of carbon in 1.00g of boron carbide is equal to the amount of carbon in 0.80g of CO2.
We can use the following formula to find out the mass of an element in a known mass of compound.
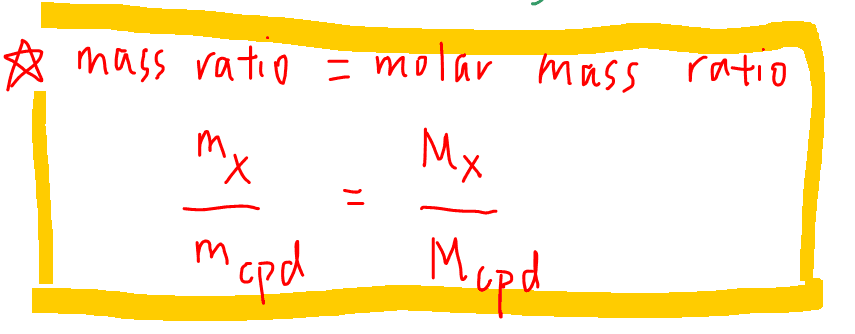
Therefore we can find the mass of boron in boron oxide and mass of carbon in carbon dioxide.
We can then calculate the moles of boron and carbon present.
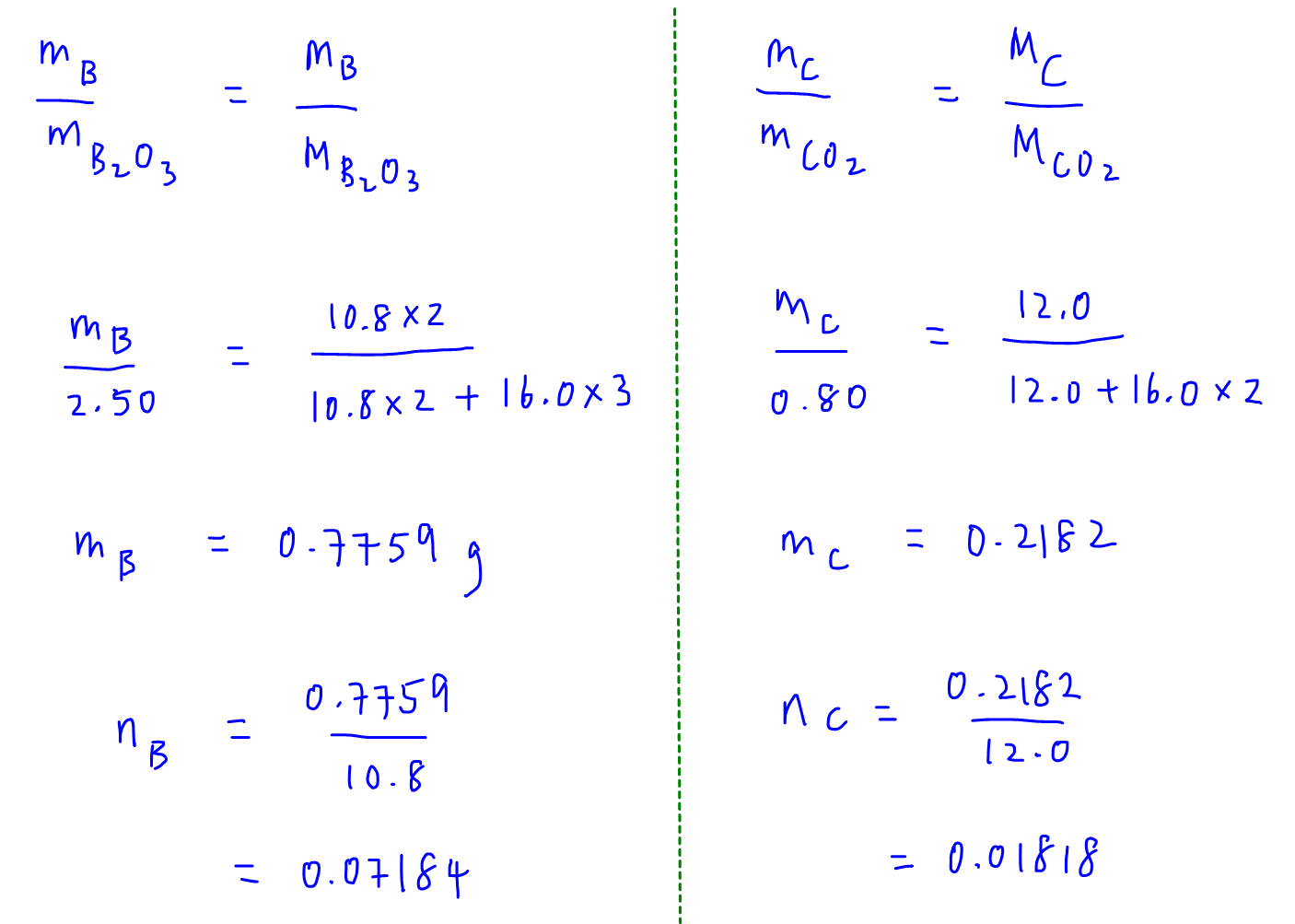
Finally we can compare the mole ratio for B to C.
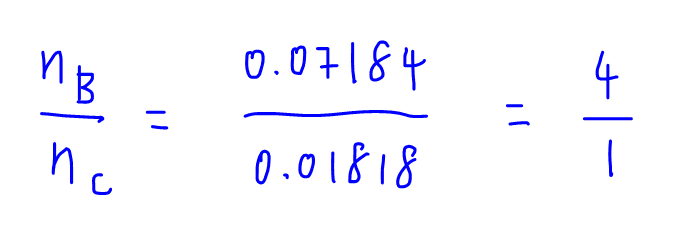
Since there are 4 B to every C, the empirical formula for boron carbide must be B4C.

So the answer to this question will be option D.
Topic: Mole Concept, Physical Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2020 A Level H2 Chemistry Paper 1.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's trusted JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
