2021 P1 Q1 - Deflection of Subatomic Particle Through Electric Field
Let Chemistry Guru, Singapore's top choice for JC Chemistry tuition, guide you through 2021 A Levels H2 Chemistry Paper 1 Question 1.
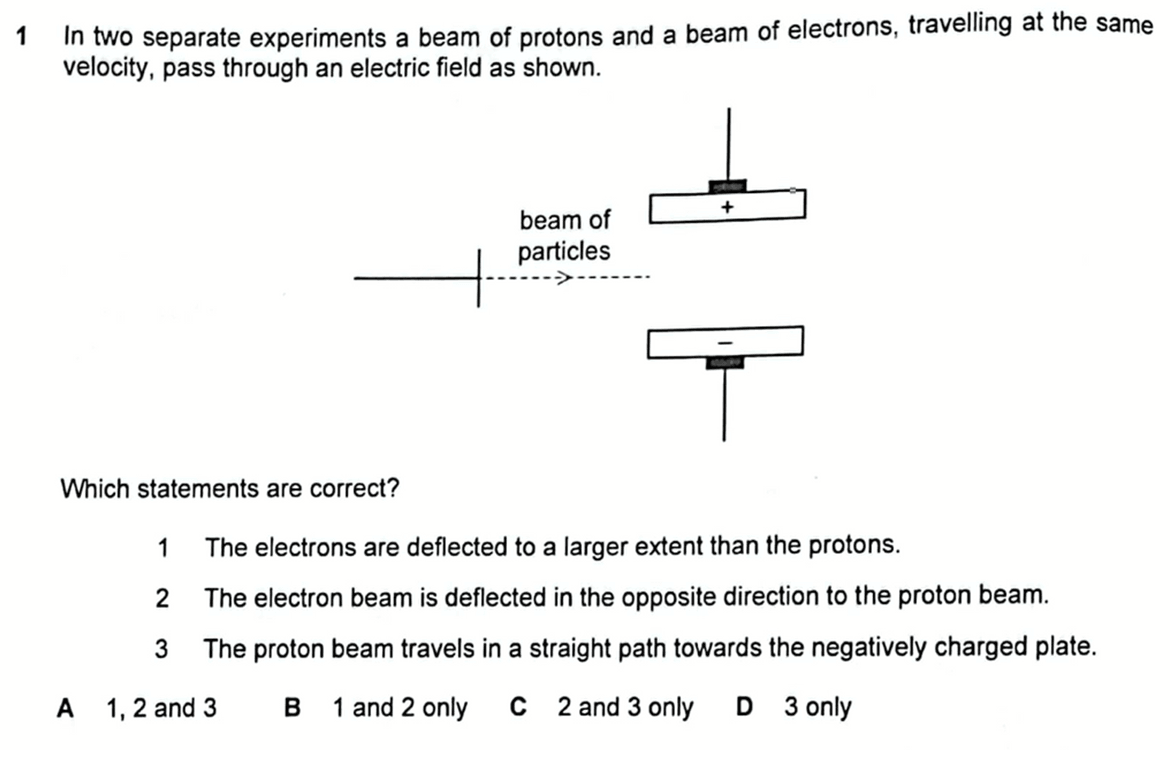
We are required to deduce if the following statements about deflection of a beam of protons and electrons through an electric field are correct.
1. The electrons are deflected to a larger extent than the protons.
The angle of deflection of a particle through an electric field is directly proportionate to its charge-mass ratio.

Since the mass of an electron is much smaller than that of a proton, its charge-mass ratio is significantly higher.
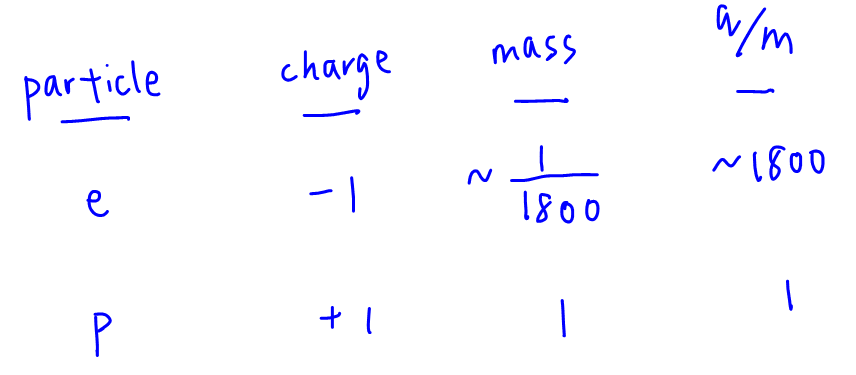
So we will expect the electron beam to be deflected to a greater extent and statement 1 is correct.
2. The electron beam is deflected in the opposite direction to the proton beam.
Since electrons and protons are oppositely charged, they should be attracted to different poles.
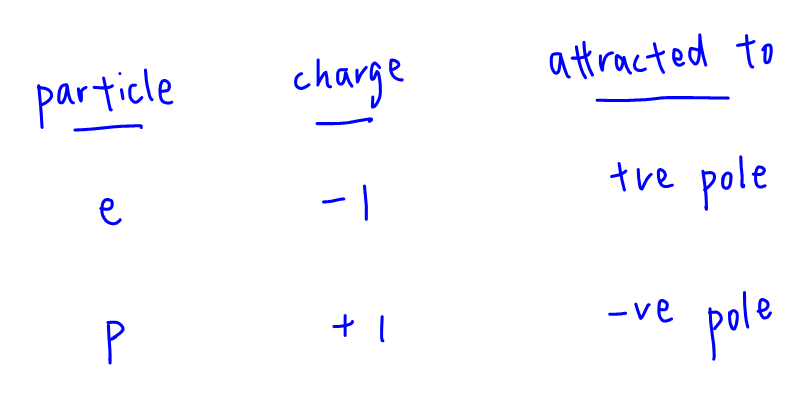
Hence statement 2 is correct.
3. The proton beam travels in a straight path towards the negatively charged plate.
Travelling in a straight path means there is constant attraction and deflection as the proton beam gets closer to the negative pole.

Travelling in a curved trajectory means there is increasing attraction and deflection as the proton beam approaches the negative electrode.
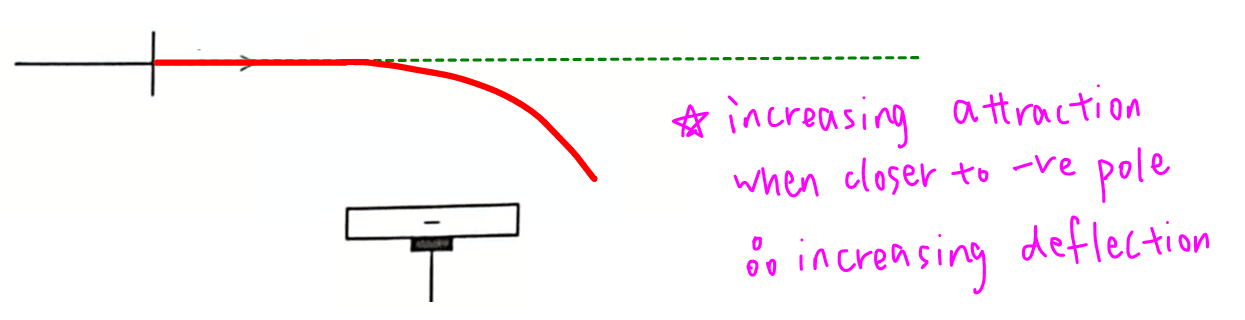
There should be a stronger attraction between the proton beam and negative pole when they are closer together hence a curved path is more reasonable.
Hence statement 3 is incorrect.
We can now compare our options and determine the answer to be option B (1 and 2 only).

Topic: Atomic Structure, Physical Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2021 A Level H2 Chemistry Paper 1.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's esteemed H2 Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
