2021 P1 Q15 - Free Radical Substitution of Methylpropane
Let Chemistry Guru, Singapore's top choice for JC Chemistry tuition, guide you through 2021 A Levels H2 Chemistry Paper 1 Question 15.

We are required to determine which of the statements about free radical substitution of methylpropane with halogen is correct.
Let's recap the mechanism of free radical substitution first.
1. Initiation
Homolytic fission of X-X bond by UV light to form halogen radicals.
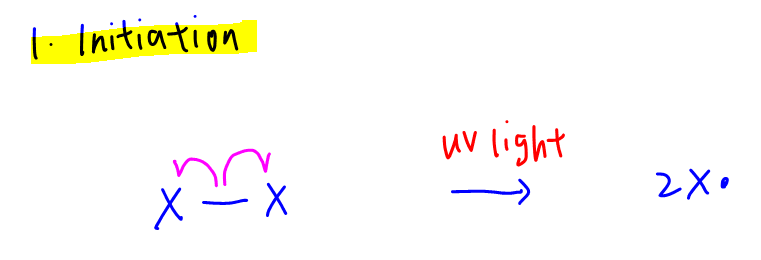
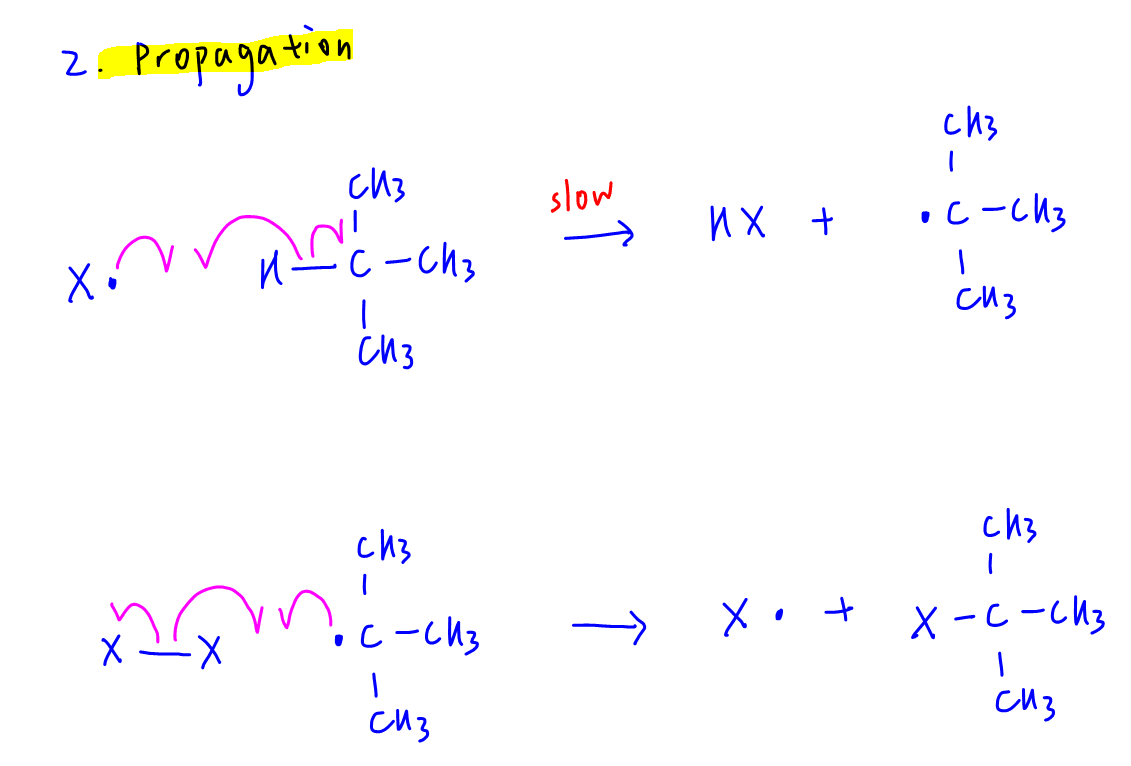
2. Propagation
X• radical attacks methylpropane to form HX and methylpropyl radical, which then attacks X2 molecule to form 2-halogenomethylpropane and regenerates X radical.
3. Termination
Homolytic fusion of two radicals to form stable compound.
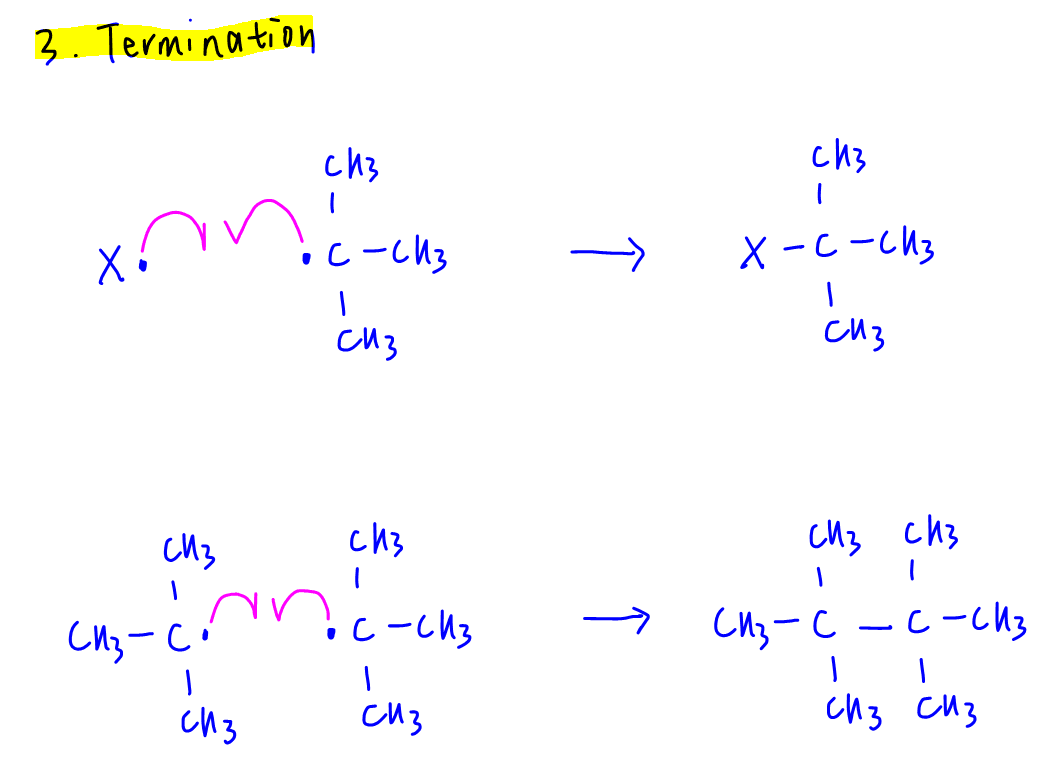
Check out the following video lesson to learn more about drawing free radical substitution mechanism.
We can now go through the options.
A - C4H9X is formed in one of the propagation reactions.
From the propagation step we can see that 2-halogenomethylpropane can be formed with molecular formula C4H9X.
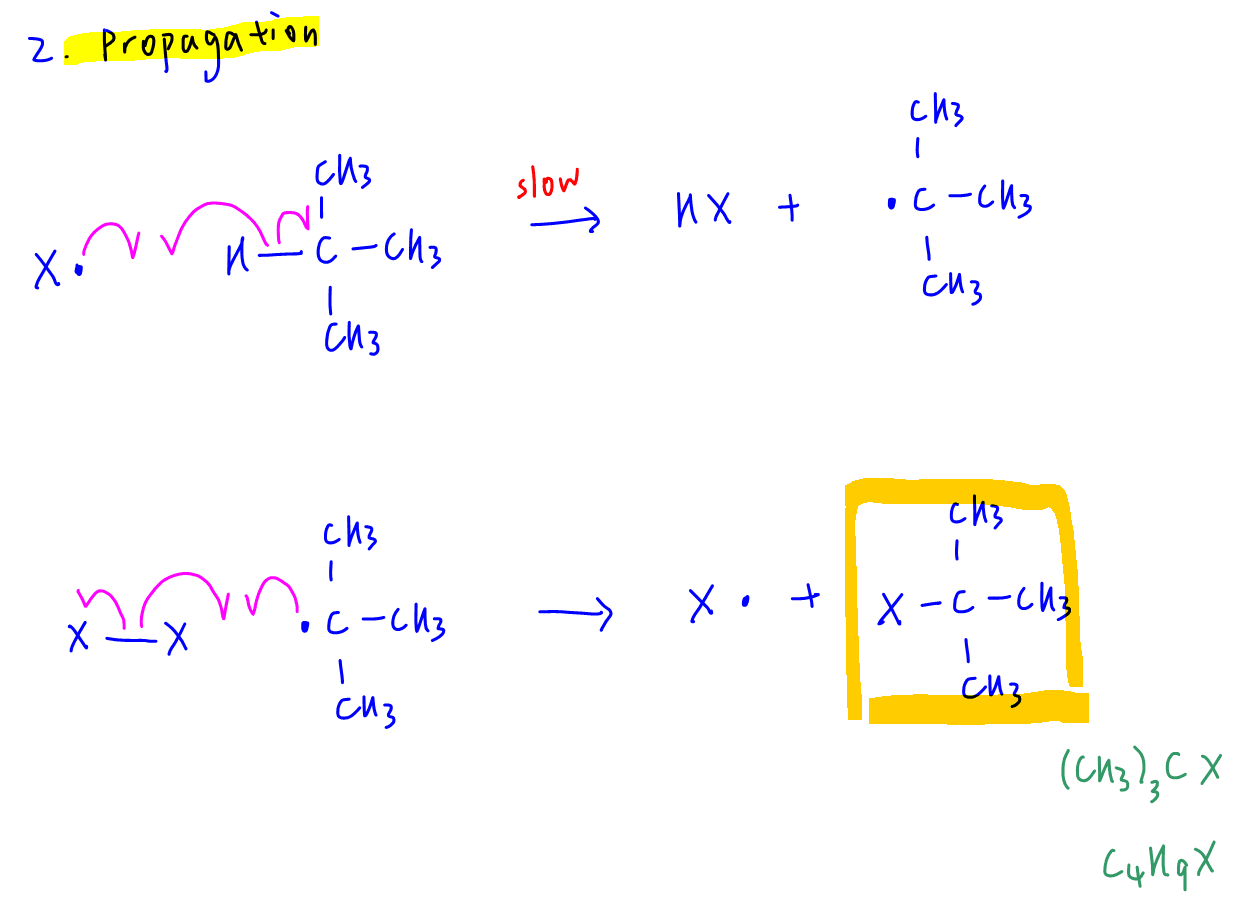
Hence statement A is correct.
B - Small amounts of (CH3)3C• are produced in a termination reaction.
The radical (CH3)3C• can be formed in the propagation step hence are found in large amounts.
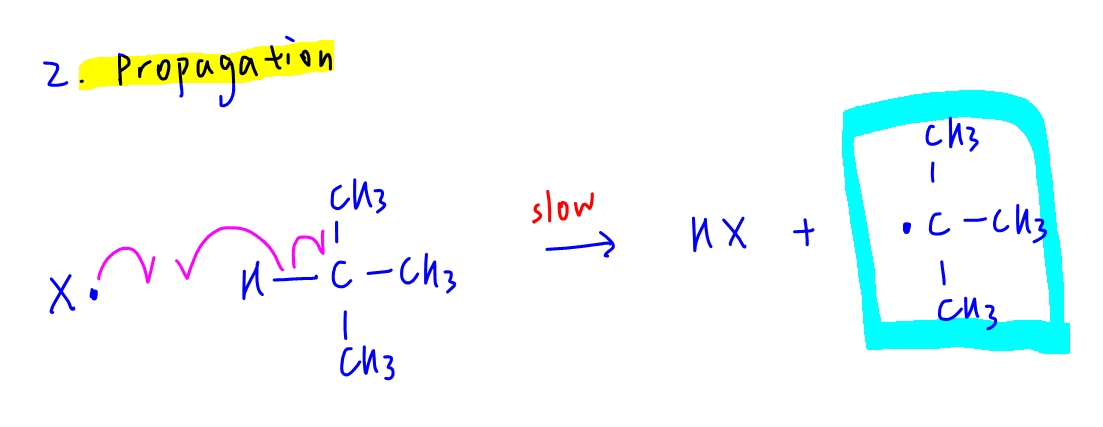
Therefore statement B is wrong.
C - The Br-Br bond is weaker than the Cl-Cl bond and so the reaction with bromine is more exothermic.
We need to calculate the enthalpy change of reaction with Br2 and Cl2.

For reaction with bromine, enthalpy change is -43 kJmol-1.
For reaction with chlorine, enthalpy change is -117 kJmol-1.
Therefore reaction with bromine is less exothermic and statement C is wrong.
D - The initiation reaction produces a halide ion which is reactive.
Free radicals instead of halides are formed in the initiation step.
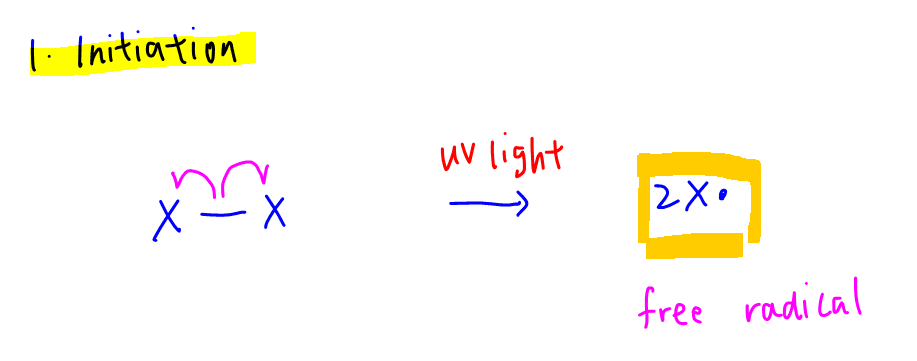
Hence statement D is wrong.
Finally we can conclude that option A is the answer to this question.

Topic: Alkanes, Organic Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2021 A Level H2 Chemistry Paper 1.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's esteemed H2 Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
