2021 P1 Q2 - Determine Group and Nucleon Number of Q
Let Chemistry Guru, Singapore's top choice for JC Chemistry tuition, guide you through 2021 A Levels H2 Chemistry Paper 1 Question 2.

We are required to determine the Group and nucleon number of element Q.
1. Determine Group

Since Q forms an ionic nitrate, Q must be a metal.
From formula QNO3, we can deduce that charge of Q is +1.
Hence Q must be a Group 1 metal.
2. Determine Nucleon Number
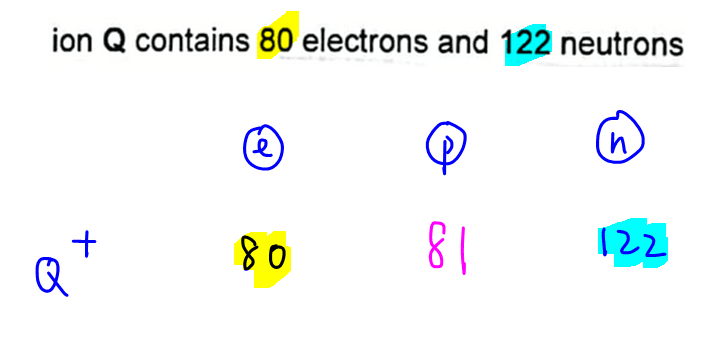
Since ion Q+ has 80 electrons, we can deduce its proton number is 81.
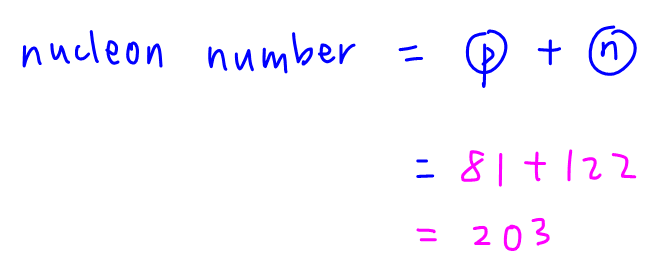
Therefore its nucleon number (proton number + neutron number) is 203.

Finally we can determine the answer to be option B. (please see below for corrected answer)
------------------------
Thank you viewer @limabashori for pointing out this discrepancy that in the Periodic Table, the element with proton number 81 is Thallium which is in Group 13.
I've assumed that since the formula of nitrate is QNO3, Q has +1 charge and only Group 1 metals can form +1 charge cations.
Usually we do not expect Group 13 metals to lose only 1 electron to form +1 charge cations.
After some research on the internet I realised that Thallium can indeed form Tl+, and its relative molecular mass of 204.4 is much closer to the calculated nucleon number of 203 that we have for this question.
So Q should be Thallium which is in Group 13 and nucleon number of 203.
Option D should be the correct answer then.
----------------------------
Topic: Atomic Structure, Physical Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2021 A Level H2 Chemistry Paper 1.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's esteemed H2 Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
