2021 P1 Q22 - Explain Why Amides are Neutral
Let Chemistry Guru, Singapore's top choice for JC Chemistry tuition, guide you through 2021 A Levels H2 Chemistry Paper 1 Question 22.

We are required to explain why an aqueous solution of N-methylethanamide has no effect on red litmus.
This means that the amide is neutral and the lone pair on nitrogen in amide is not donated.
We can draw resonance structures of amide to show that the lone pair on N is delocalised into the acid group.
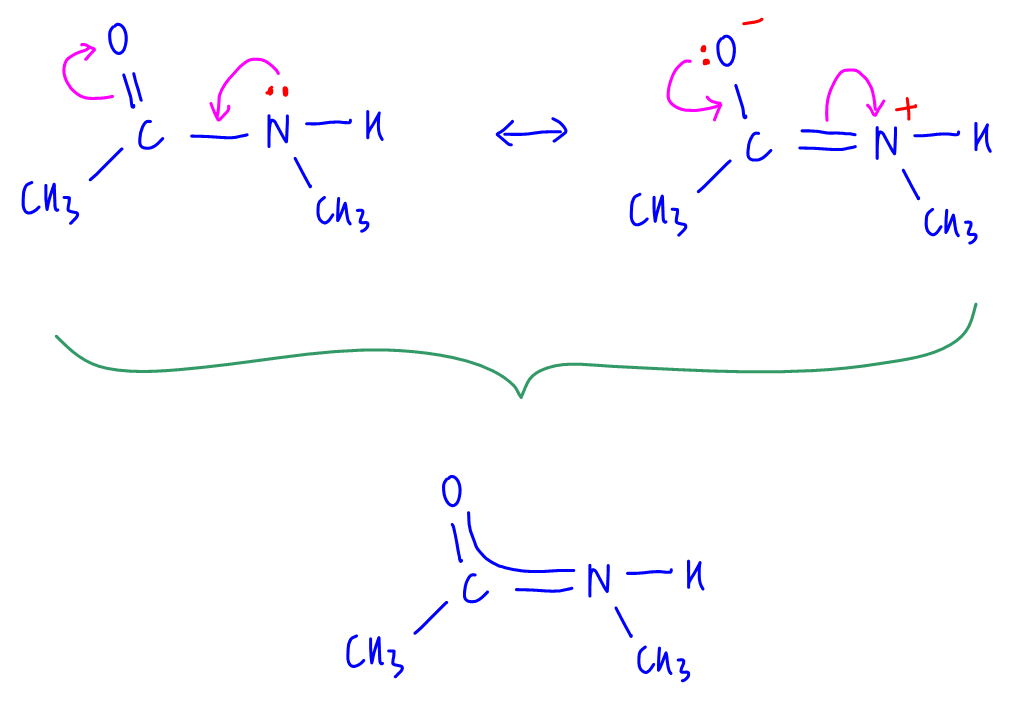
The lone pair on nitrogen is delocalised extensively between electronegative N and O and amide functional group is stabilised by resonance.
The lone pair will not be used for donation since this will result in loss of resonance stability.
Hence amides are neutral and will not give any observation with red litmus.
We can now go through the statements and see which are relevant in explaining the neutrality of amides.
1. Delocalisation of the lone pair of electrons on the nitrogen atom over the C=O group results in the nitrogen becoming less available for protonation.

This is exactly what we've explained earlier and the lone pair will not be used for protonation to maintain the resonance stability in amide functional group.
Hence statement 1 explains why amide is neutral.
2. Delocalisation of the lone pair of electrons on the nitrogen atom over the C=O group strengthens the C-N bond.
This is true since the delocalisation of lone pair is over oxygen, carbon and nitrogen atoms hence C-N bond is stabilised and strengthened by resonance.
However the C-N bond is not broken during neutralisation so its strength has no effect on the neutrality of amides.
Therefore statement 2 does not explain why amide is neutral.
3. N-methylethanamide is more stable than the ion that forms when it is protonated.
We can look at the protonation of amide and compare the stability of amide and conjugate acid formed.

Reactant amide is resonance stabilised while product conjugate acid has no resonance stability so is significantly less stable.
The position of equilibrium will lie significantly towards the left hand side to favour the formation of more stable amide.
This means amide will not have the tendency to accept proton hence it is neutral.
Hence statement 3 explains why amide is neutral.
We can now go through the solutions and determine the answer to this question is option A.

Topic: Nitrogen Compounds and Proteins, Organic Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2021 A Level H2 Chemistry Paper 1.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's top JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
