2021 P1 Q3 - Comparing Ionisation Energies of Different Elements
Let Chemistry Guru, Singapore's top choice for JC Chemistry tuition, guide you through 2021 A Levels H2 Chemistry Paper 1 Question 3.
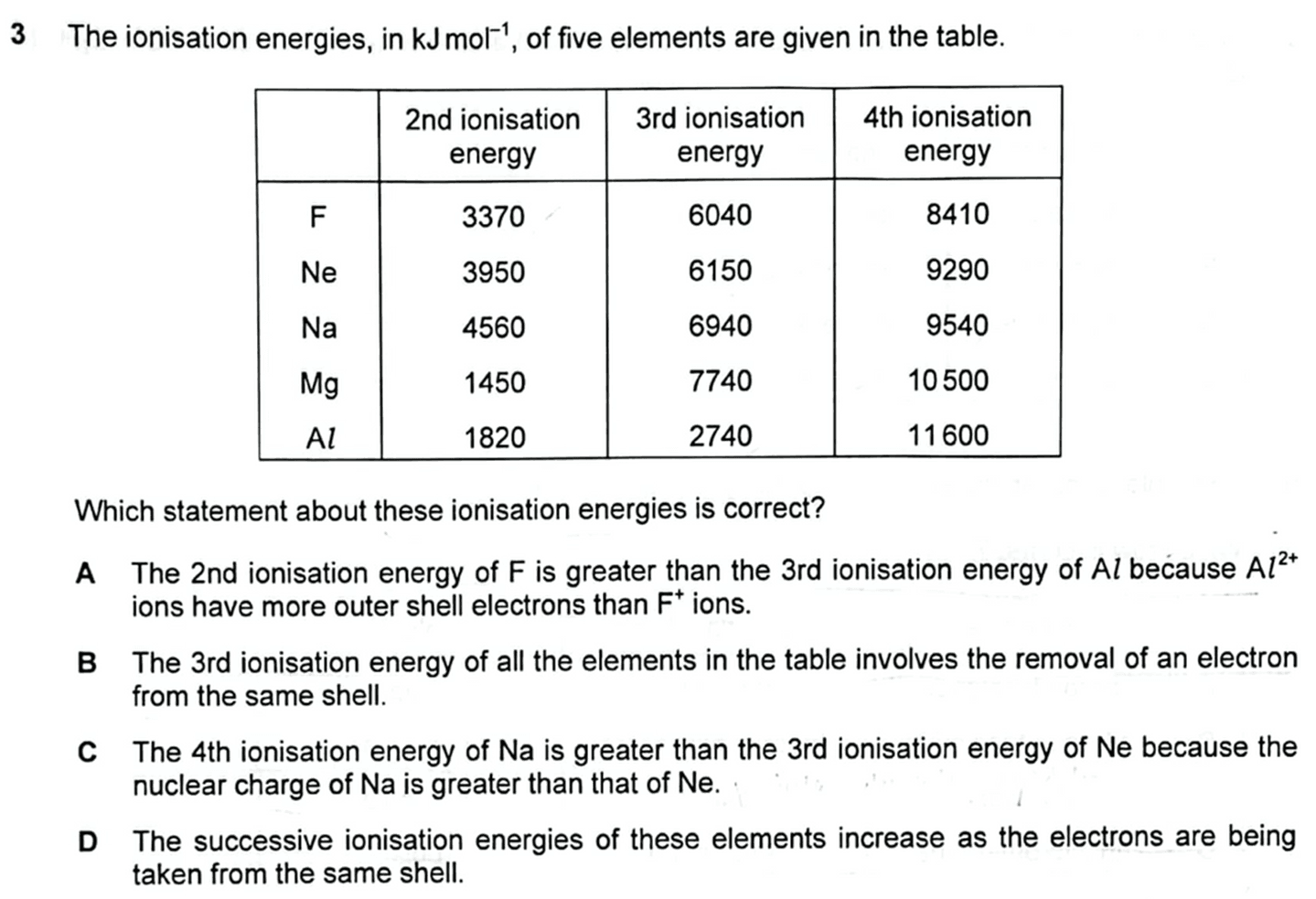
We need to determine which of the statements about ionisation energies is correct.
First let us write down the equations that correspond to second, third and fourth ionisation energies:
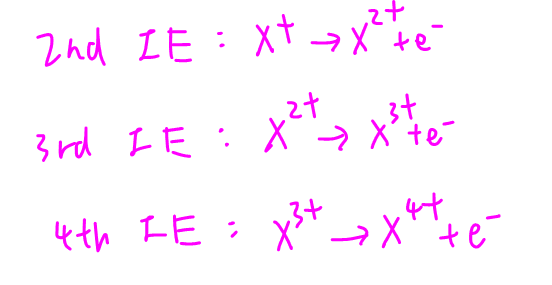
Notice second IE is removing electron from X+ ion, third IE is removing electron from X2+ ion and fourth IE is removing electron from X3+ ion.
Next let us also write out the electronic configurations for elements F to Al so that it will be easier to deduce the electronic configurations for various ions when we run through the statements.

Statement A: 2nd IE of F is greater than 3rd IE of Al because Al2+ ions have more outer shell electrons thatn F+ ions.
2nd IE of F is removing electron from F+ while 3rd IE of Al is removing electron from Al2+

F+ has 6 valence electrons from second shell while Al2+ has 1 valence electron from third shell.
Hence statement A is incorrect.
2nd IE of F is greater as electrons are removed from second principal quantum shell which is much closer to the nucleus hence more energy is required to overcome the stronger attraction between the nucleus and valence electron.
Statement B: 3rd IE of all the elements in the table involves the removal of an electron from the same shell.
Here's the list of 3rd IE for all the elements:

Electron of Al2+ is removed from third shell while the other +2 charge cations have their electrons removed from second shell.
Hence statement B is incorrect.
Statement C: 4th IE of Na is greater than 3rd IE of Ne because the nuclear charge of Na is greater than that of Ne.
Comparing 4th IE of Na and 3rd IE of Ne:

Both ions are isoelectronic hence will have the same shielding effect.
Na3+ has more protons and greater nuclear charge than Ne2+ while shielding effect is the same since Na3+ and Ne2+ are isoelectronic.
Na3+ has greater effective nuclear charge, stronger attraction between nucleus and valence electron, need more energy to remove electron and hence 4th IE of Na is greater.

Therefore statement C is correct.
Statement D: Successive IE of these elements increase as the electrons are being taken from the same shell.
Comparing 2nd to 4th IE of all elements we notice there is a change in principal quantum shell from 2nd to 3rd IE of Mg, and 3rd to 4th IE of Al.

Hence statement D is incorrect.
We can now compare our options and determine the answer to this question is statement C.

Topic: Atomic Structure, Physical Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2021 A Level H2 Chemistry Paper 1.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's esteemed H2 Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
