A Level Chemistry 2017 Paper 1 Question 23 Solution - Exclusive
In this exclusive video we want to discuss the suggested solution for A Levels Chemistry (H2 Chemistry) 2017 Paper 1 Question 23.
Let Chemistry Guru, Singapore's top JC Chemistry tuition centre, guide you through this question:
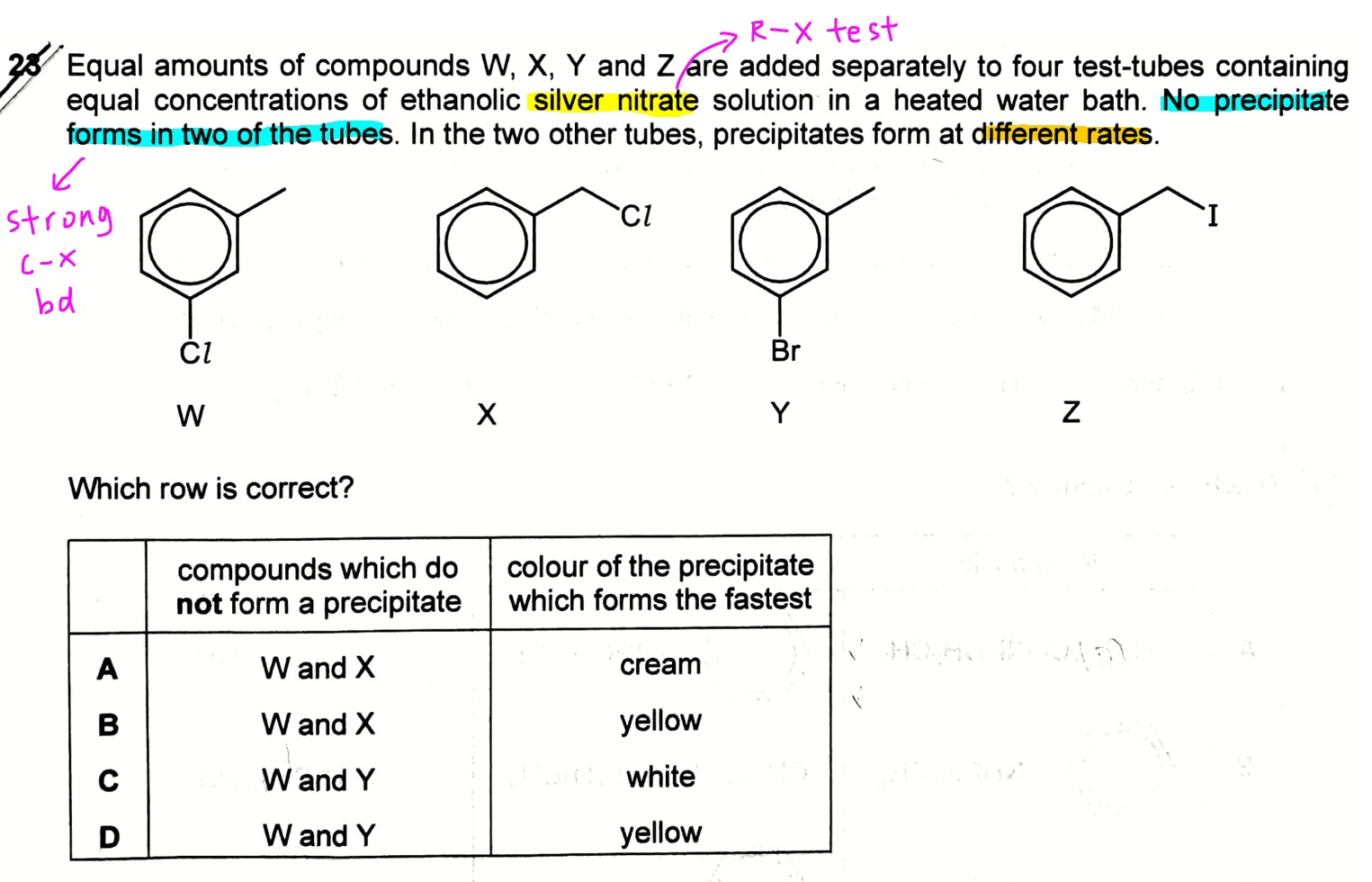
The concept tested in this question is on the reactivity of C-X bonds in Halogenoalkanes.
For a list of all the reactions of halogenalkanes, do check out this video lesson.
When silver nitrate is used in organic chemistry questions, we are testing for the presence of sliver halide precipitates, where the halides are released from nucleophilic substitution reactions of halogenoalkanes or alkylhalides.
Observation 1 - No precipitate formed in 2 of the tubes
When no precipitate is formed, it means the C-X bond is stable and resistant to nucleophilic substitution.
Halogenobenzene will give this observation as the lone pair of halogen can interact with the delocalised pi system of benzene and the C-X bond is stablised by resonance which makes it unreactive.
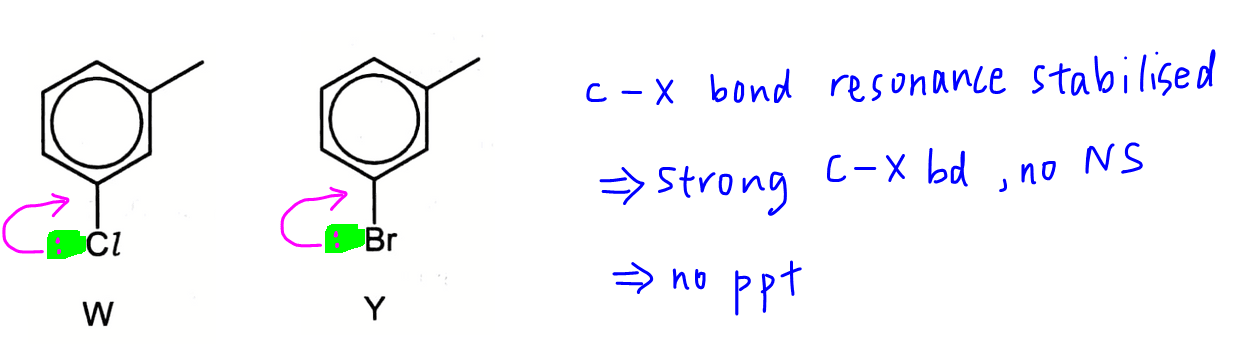
Hence compounds W and Y will not form a precipitate, and options A and B are eliminated.

Observation 2 - Precipitate formed at different speeds
Comparing choloroalkane X and iodoalkane Z, the C-I bond in Z is longer and weaker hence is more easily broken to release I- for precipitation.

This means the formation of yellow AgI precipitate will be faster as compared to the formation of white AgCl precipitate, and option C is eliminated.

Therefore the answer to this question will be option D.
Check out this video for the full solution and detailed explanation!
Topic: Halogenoalkanes, Organic Chemistry, A Level Chemistry, Singapore
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's leading JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
You might also be interested in the suggested solution for Paper 1 Question 22.
Check out other A Level Chemistry Video Lessons here!
Looking for H2 Chemistry Tuition? Do consider taking up my classes at Bishan or online classes!
