A Level Chemistry 2017 Paper 1 Question 27 Solution - Exclusive
In this exclusive video we want to discuss the suggested solution for A Levels Chemistry (H2 Chemistry) 2017 Paper 1 Question 27.
Let Chemistry Guru, Singapore's top JC Chemistry tuition centre, guide you through this question:

The topic tested in this question is on SN1 Mechanism of Halogenoalkane.
Since the solvent has to be polar, we can immediately eliminate option B since C6H14 is non-polar.
From the second step of the mechanism, OH- from NaOH acts as a nucleophile and attack the carbocation to form an alcohol product.

A by-product can be formed in the second step when the polar solvent acts as a competing nucleophile to attack the carbocation.
Hence we need to consider the nucleophilic attack for each polar solvent and determine if a by-product is formed.
A. H2O solvent
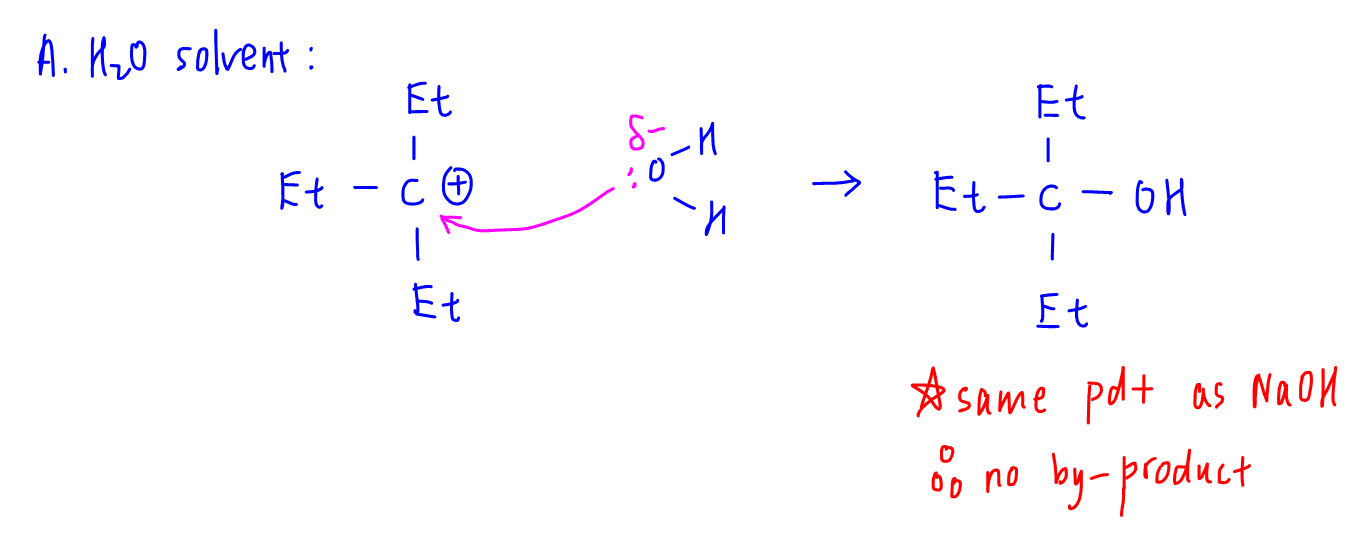
On nucleophilic attack of H2O and subsequent deprotonation, the product formed is an alcohol which is the same product formed from NaOH.
Therefore there are no by-products and option A is the most likely answer.
C. CH3CH2NH2 solvent

On nucleophilic attack of amine CH3CH2NH2 and subsequent deprotonation, produced formed is a secondary amine.
A different by-product is formed.
D. CH3CH2OH solvent

On nucleophilic attack of alcohol CH3CH2OH and subsequent deprotonation, produced formed is an ether.
A different by-product is also formed.
Finally we can compare the options and confirm the answer to this question is option A.

Check out this video for the full solution and detailed explanation!
Topic: Halogenoalkanes, Organic Chemistry, A Level Chemistry, Singapore
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's reputable A Level Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
You might also be interested in this video on A Level Chemistry 2017 Paper 1 Question 26 Solution.
Check out other A Level Chemistry Video Lessons here!
Looking for H2 Chemistry Tuition? Do consider taking up my classes at Bishan or online classes!
