Aldol Addition Reaction
In this video created by Chemistry Guru, Singapore's leading JC Chemistry tuition centre, let's discuss aldol addition reaction of carbonyl compounds.
The name aldol is derived from aldehyde (ald-) and alcohol (-ol), but compounds which contain ketone and alcohol are also considered as aldols.
This reaction is not in A Level Chemistry Syllabus but is quite common in JC Prelim questions so it's a good idea to have an idea what this reaction is about.
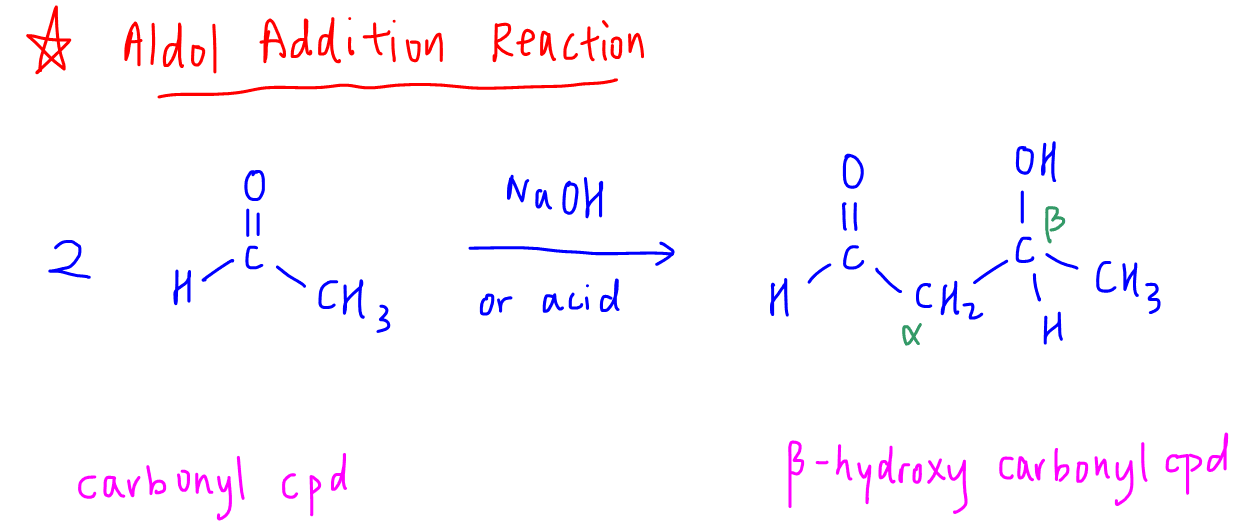
Using a strong acid or base catalyst, 2 carbonyl compounds can react to form a beta-hydroxy carbonyl compound.
The alcohol functional group is 2 carbons away from the carbonyl functional group.
Let's consider the mechanism of aldol addition reaction, using a base catalyst as an example, to understand this better.
Mechanism (base catalysed)
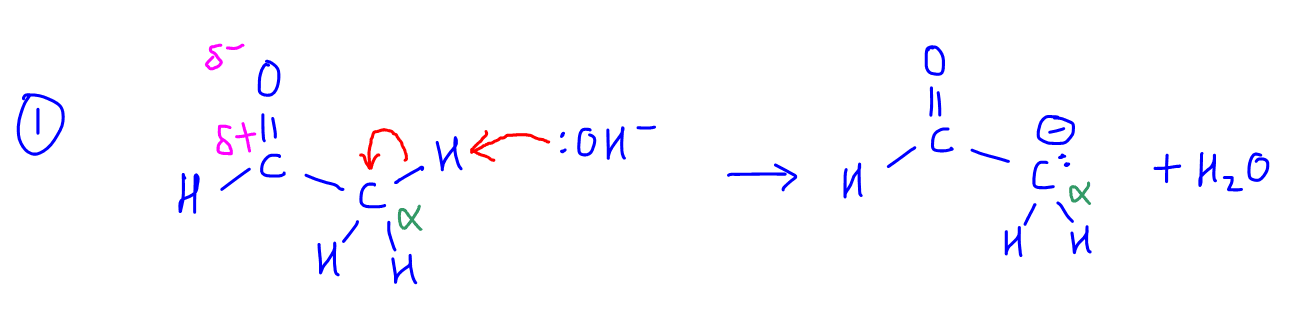
1. The base can extract a proton from alpha-alkyl carbon to form a negatively charged alpha carbanion.
2. The carbonanion acts as nucleophile and attack carbonyl carbon.
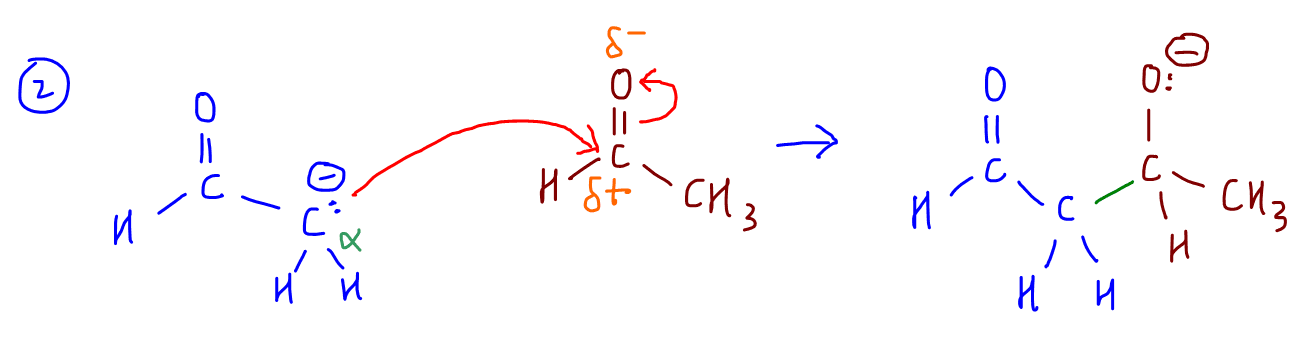
Notice this is very similar to the nucleophilic addition reaction that is in A Level Chemistry syllabus.
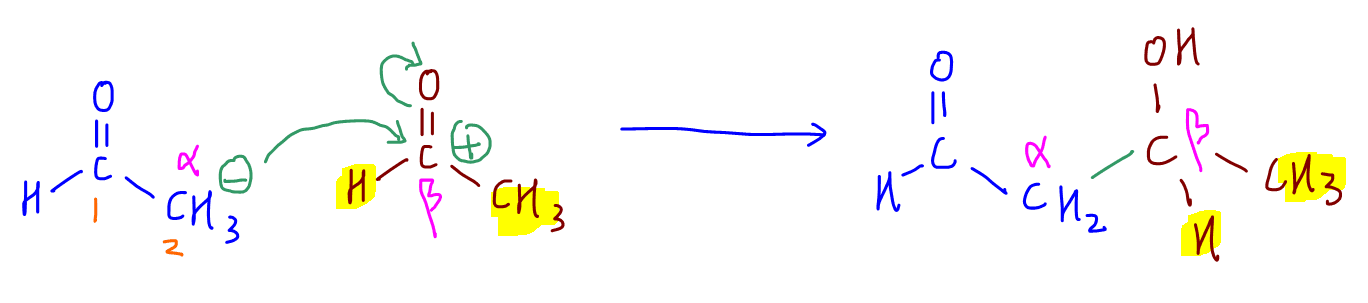
3. The negative oxygen extracts a proton from water molecule to form the product and regenerates OH- catalyst

Predicting Products for Aldol Addition Reaction
With the understanding of the mechanism we can now predict the products that each of these carbonyl compounds can form when they undergo aldol addition reactions.
1. ethanal
2. propanone
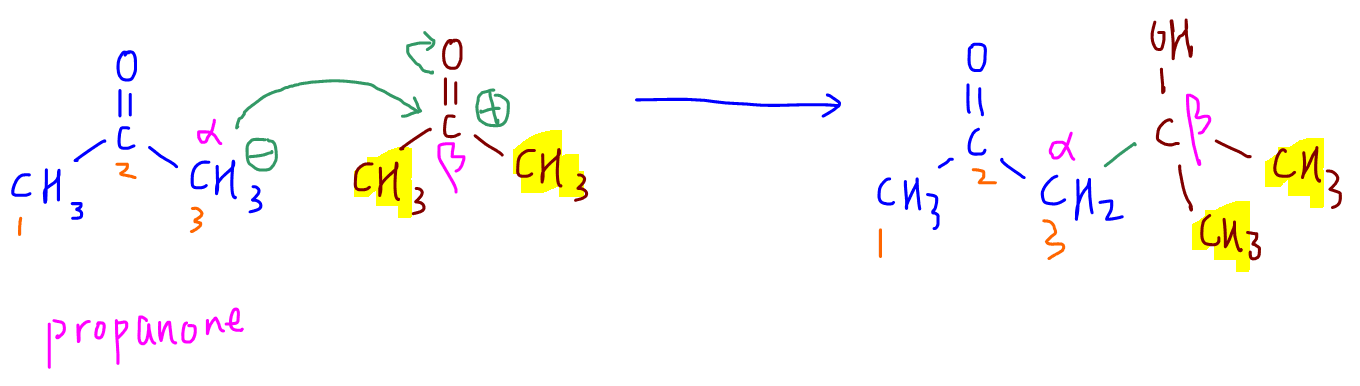
3. butanone
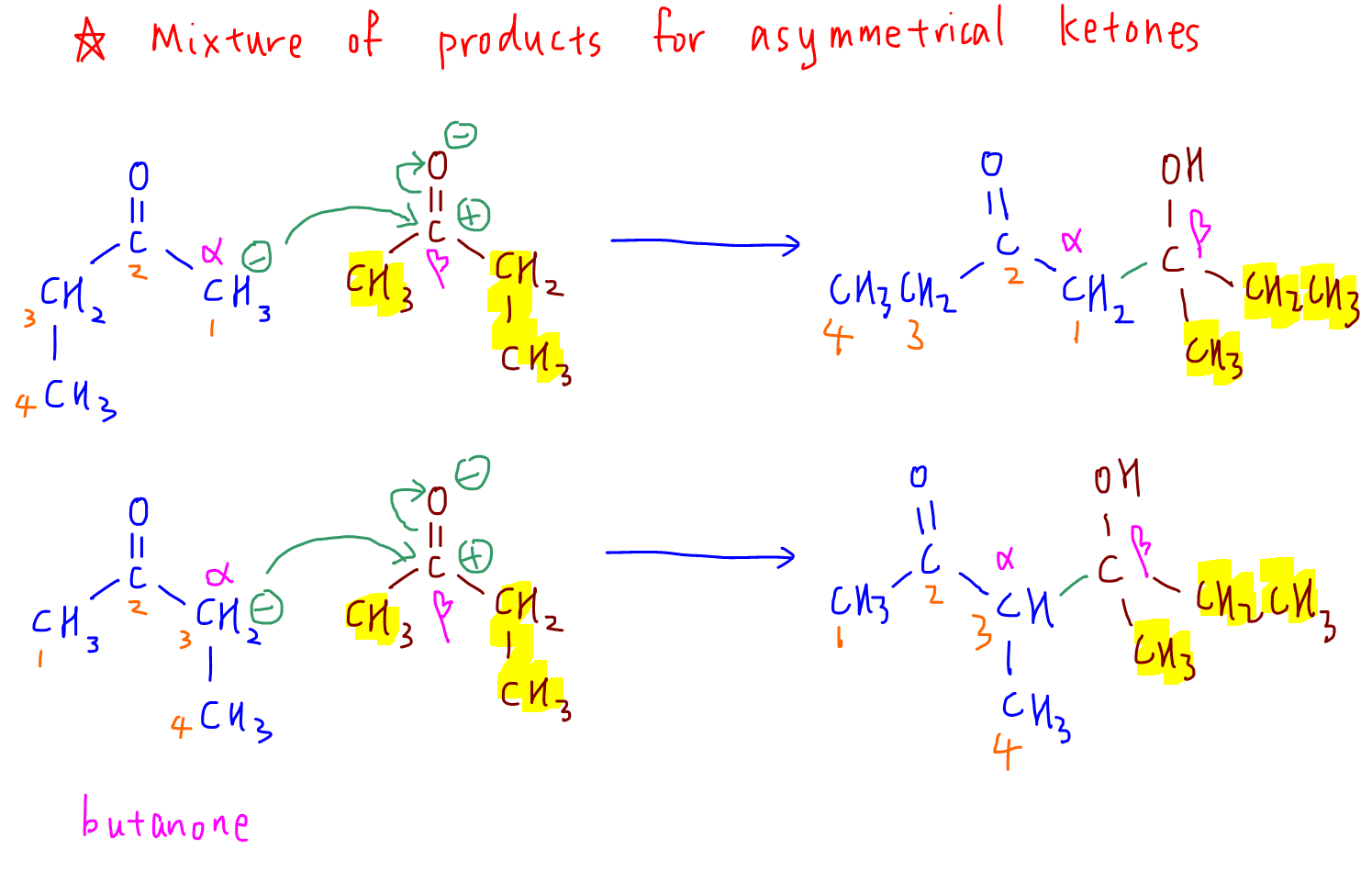
Notice when the ketone is asymmetrical we can have a mixture of products.
In fact we can have 2 different carbonyl compounds reacting together via aldol addition to give many different products.
To figure out the products, watch the step-by-step discussion in this video!
Topic: Carbonyl Compounds, Organic Chemistry, A Level Chemistry, Singapore
Back to other previous Organic Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's renowned A Level Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my A Level H2 Chemistry Tuition classes at Bishan or online tuition classes!
