How to Calculate pH at Equivalence Point
In this JC2 webinar we want to learn how to calculate the pH at equivalence point.
Question: Determine the pH of resultant solution when 25.0 cm3 of 0.1 moldm-3 NaOH is added to 25.0 cm3 of 0.1 moldm-3 CH3COOH. Assume Ka of CH3COOH is 10-5 moldm-3.
The ICE table is very useful to determine the resultant solution of an acid-base reaction.
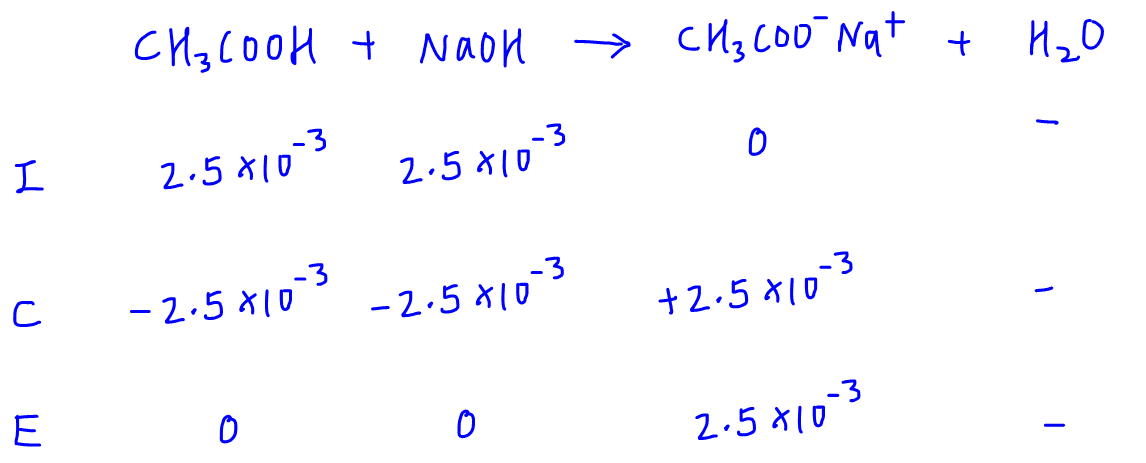
The resultant solution at complete neutralisation is the salt solution of CH3COONa.
Therefore the pH at equivalence point will just be the pH of this salt solution.
We need the concept of salt hydrolysis to deduce the nature of this salt.
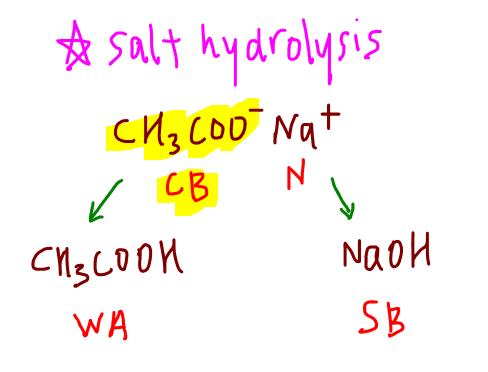
If you are interested to know about salt hydrolysis in detail, check out this video that I've previously done on how to deduce the nature of salts.
Sodium ethanoate is alkaline since CH3COO- is the conjugate base of weak acid CH3COOH.
Hence it will dissociate in water to give OH- and make the solution alkaline.

Now that we know the equivalence point is alkaline, how do we calculate its pH?
Notice we have established the following relationship:
equivalence point → salt → conjugate base → weak base
Therefore we can use the formula of weak base to calculate the OH- concentration at this equivalence point.
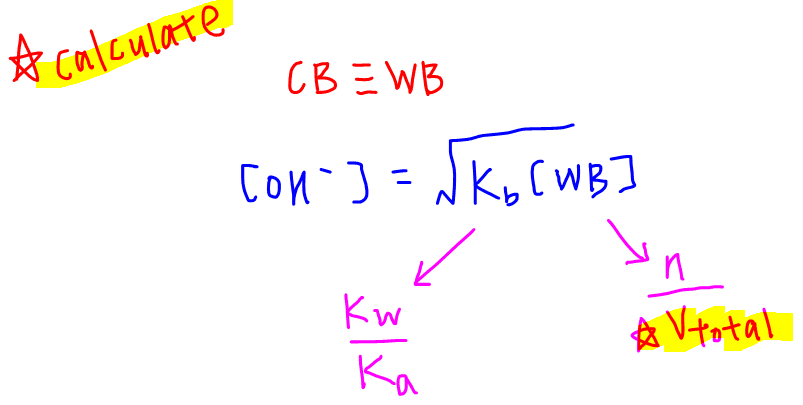
Kb is not given but we can calculate it easily from ionic product of water Kw and Ka via the formula Kw = Ka.Kb

Concentration of CH3COO- can also be easily calculated, keeping in mind the total volume is 50cm3.

We can now substitute these back into the weak base formula to find OH- concentration, and eventually the pH at this equivalence point.
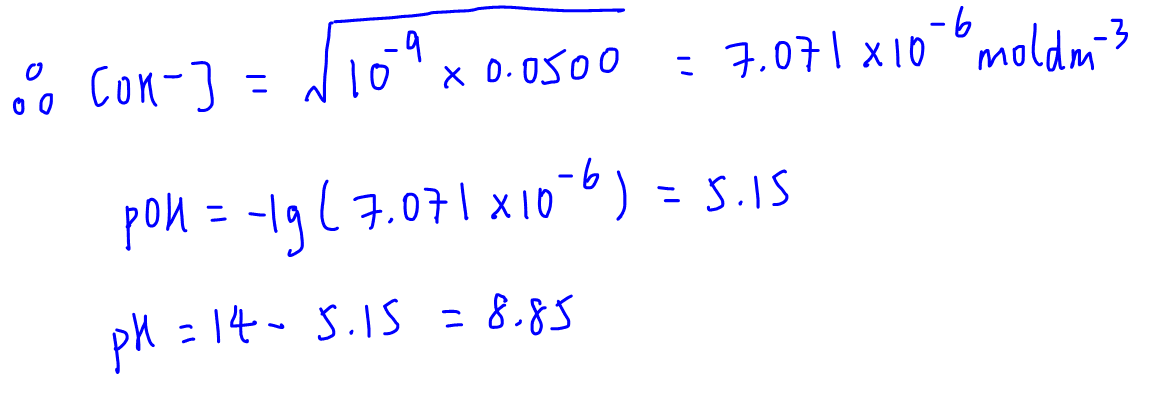
Topic: Buffer Solution and Titration Curve, Physical Chemistry, A Level Chemistry, Singapore
Back to other previous Physical Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's trusted JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
