2019 A Level H2 Chemistry Paper 1 Question 30 - Calculate Time Taken for Copper Electrolysis
We are finally at our last question (Q30) for 2019 A Level H2 Chemistry Paper 1!
Let Chemistry Guru, Singapore's renowned JC Chemistry tuition centre, guide you through the question.

We need to calculate the time taken to cover both sides of the copper with new copper to a total depth of 1000 atoms.
First let's determine the number of copper atoms per layer.
Given the dimensions of the square copper cathode is 0.1m x 0.1m and of each copper atom is 3.0 x 10-12m x 3.0 x 10-12m.

Hence the number of copper atoms per layer will be 1.111 x 1021
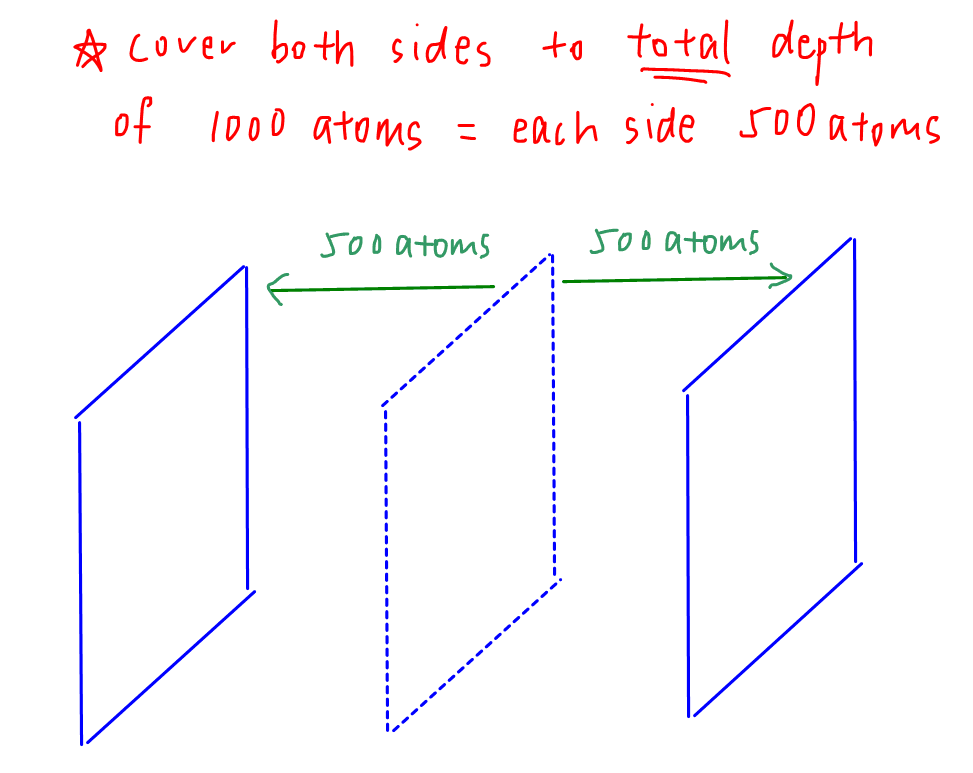
Next we have to interpret the statement "cover both sides of the piece of copper with new copper to a total depth of 1000 atoms".
Many students see this as a depth of 1000 atoms to the lefthand side plus a depth of another 1000 atoms to the righthand side to give a total depth of 2000 atoms.
However since the question states a "total depth of 1000 atoms", we should interpret it as a depth of 500 atoms to the LHS plus a depth of another 500 atoms to the RHS to give a total depth of 1000 atoms.
We can now find the total number of copper atoms and then the moles of copper discharged will be 1.845 mol.

Next we can work out the moles of electron involved in the electrolysis to be 3.69 mol using the half equation for discharge of Cu2+ to Cu metal.

Finally we can use both Faraday's equations (Q = n.F and Q = I.t) to find time taken for the electrolysis to be 24.7 hours.

Hence the answer to this question will be option B.
This was a pretty controversial question as many students who interpreted the total depth to be 2000 atoms (as discussed earlier) will get option C as their answer.
In my opinion this question was not very well crafted as challenging questions should be set based on chemistry concepts and not due to ambiguous statements.
Topic: Electrochemistry, Physical Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2019 A Level H2 Chemistry Paper 1
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's trusted JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or online tuition classes!
