Common Ion Effect
In this JC2 webinar let's discuss common ion effect and how it suppresses solubility of sparingly soluble salts.
Let's consider the scenario where we have a solution of silver nitrate (AgNO3) and silver chloride (AgCl).
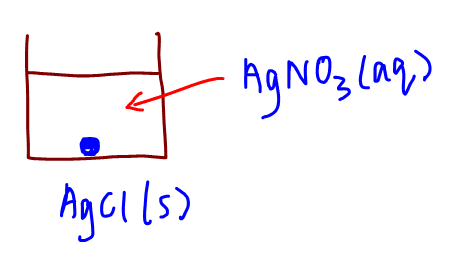
Silver nitrate is soluble so will be fully dissociated in solution to form Ag+ and NO3- ions.
Silver chloride is sparingly soluble so will only be partially dissociated to form some Ag+ and Cl- ions.
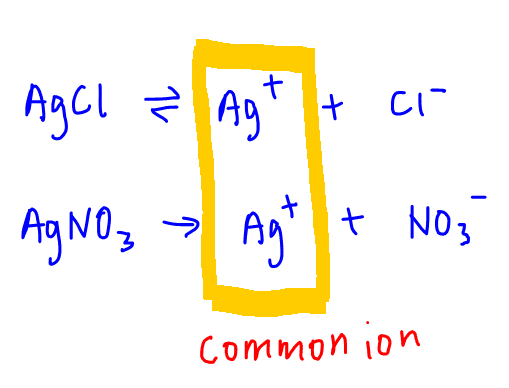
Notice both salts give Ag+ ions.
Since this ion is dissociated from both salts, we call this the common ion.
The presence of common ion from AgNO3 will increase the total Ag+ concentration in solution.
This will affect the position of equilibrium for AgCl dissociation.
According to Le Chatelier's Principle, an increase in concentration of product will shift the position of equilibrium towards the left to reduce the concentration of product.
Check out the following video lesson for a detailed discussion on Le Chatelier's Principle.
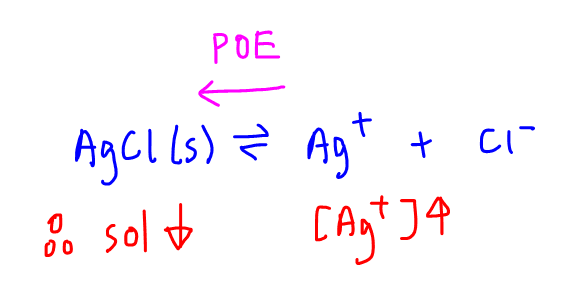
This means that more AgCl solid will be formed, and less of the salt dissolves, ie decrease or suppression of solubility.
In general the presence of a common ion will suppress the solubility of a sparingly soluble salt.
Topic: Solubility Product, Physical Chemistry, A Level Chemistry, Singapore
Back to other previous Physical Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's trusted JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
