Compare Boiling Point of Isomers
Let's check out this question.
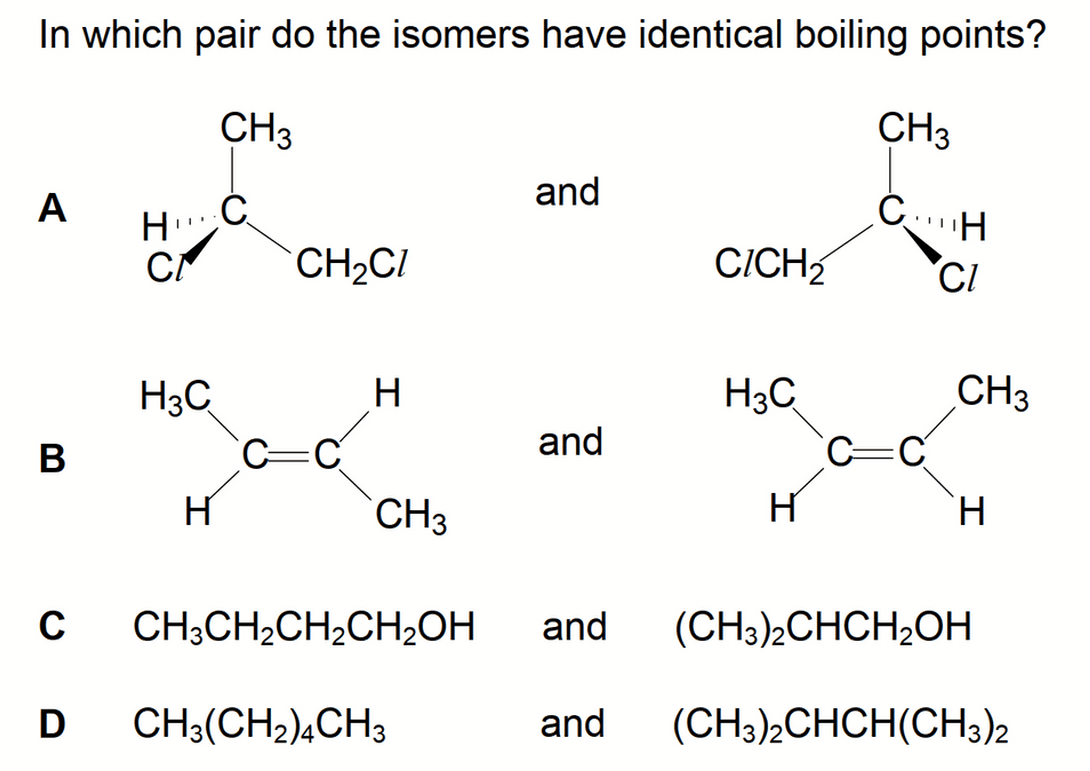
We are required to determine the pair of isomers with identical boiling points.
Let's consider each pair of isomers.
Option A - Optical Isomers or Enantiomers
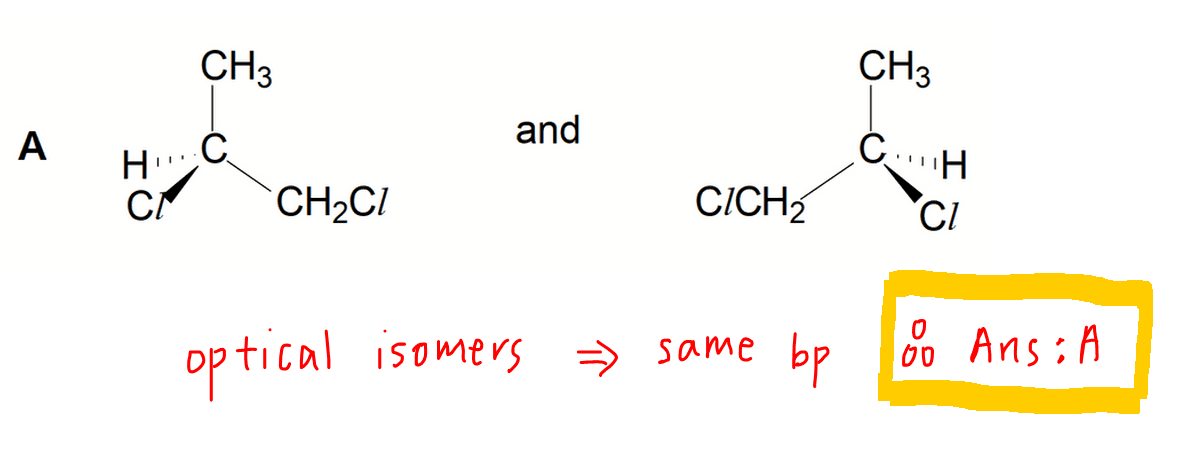
In A Level Chemistry Syllabus only optical isomers have identical boiling points.
So the answer to this question is option A.
The only difference in physical property between optical isomers is the direction of rotation of plane polarised light, or their optical activity.
Option B - Cis Trans Isomers or Geometric Isomers
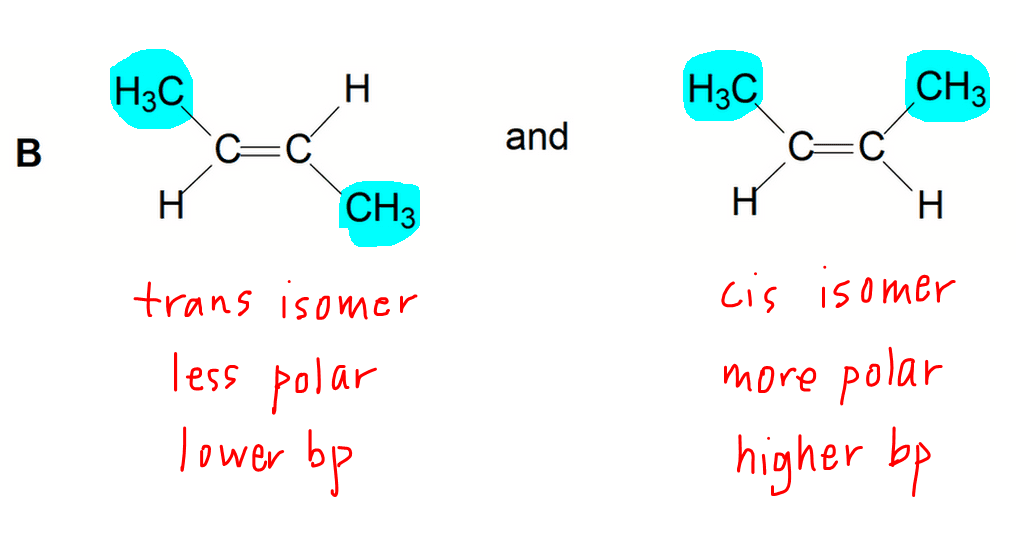
The bulky groups for cis isomers are on the same side of the alkene.
This makes the cis isomer less symmetrical and the electron cloud distribution is more unequal.
Therefore the cis isomer is more polar than the trans isomer and has a slight permanent dipole - permanent dipole attraction between molecules.
Thus the cis isomer has a higher boiling point than the trans isomer.
Interestingly the cis isomer has a lower melting point than the trans isomer, even though it is more polar.
The less symmetrical cis isomer is less efficiently packed in the solid state.
Thus the cis isomers are farther apart in the solid state, attraction between molecules are weaker, hence melting point is lower.
Option C & D - Chain Isomers
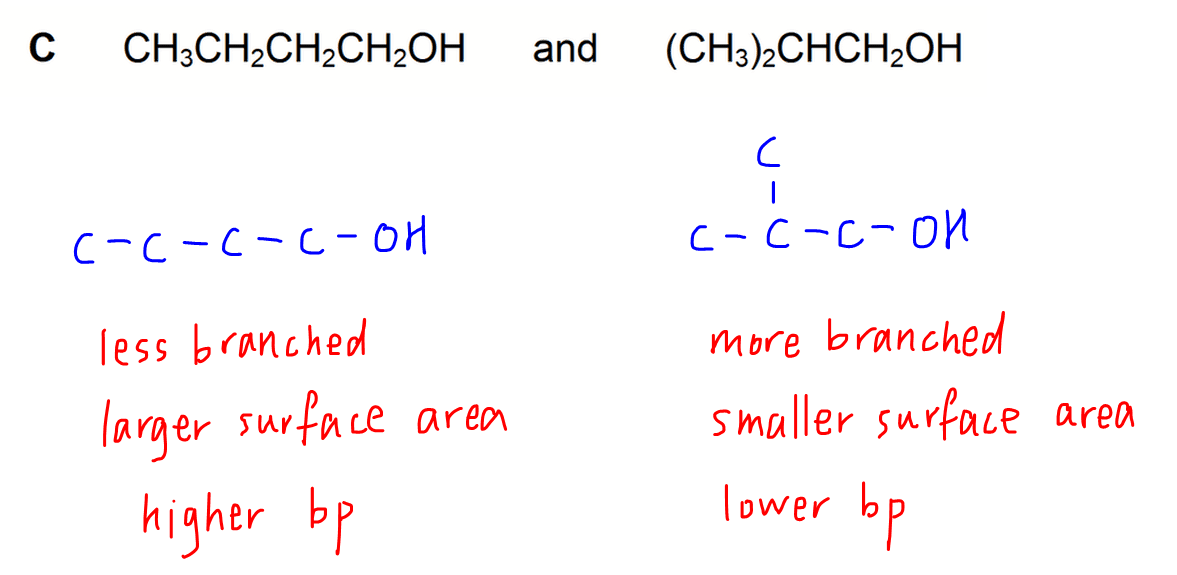
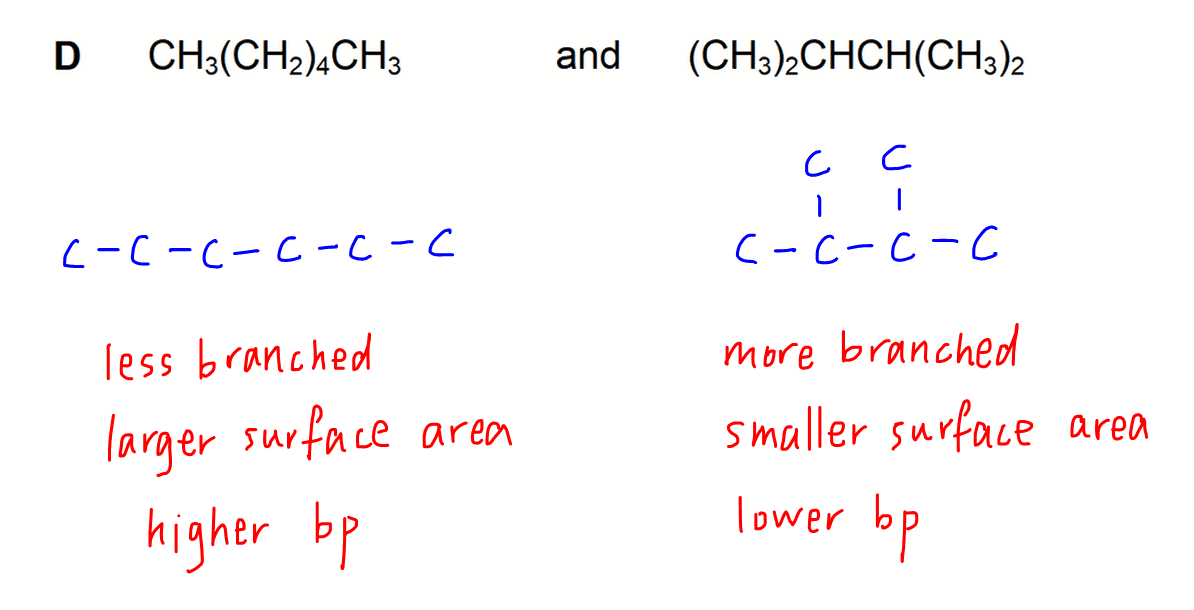
For both options the difference is in the extent of branching of carbon chain.
For the unbranched or straight chain isomer, its surface area will be larger for interaction between molecules.
Hence intermolecular attraction is stronger and boiling point is higher for the unbranched isomer.
The branched isomer will have a smaller surface area for molecular interaction which results in lower boiling point.
Topic: Introduction to Organic Chemistry, Organic Chemistry, A Level Chemistry, Singapore
Back to other previous Organic Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's top H2 Chemistry tuition choice since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or online tuition classes!
