2019 A Level H2 Chemistry Paper 1 Question 28 - Deduce Products of Hydrolysis by Chymotrypsin
Here's question 28 for 2019 A Level H2 Chemistry Paper 1.
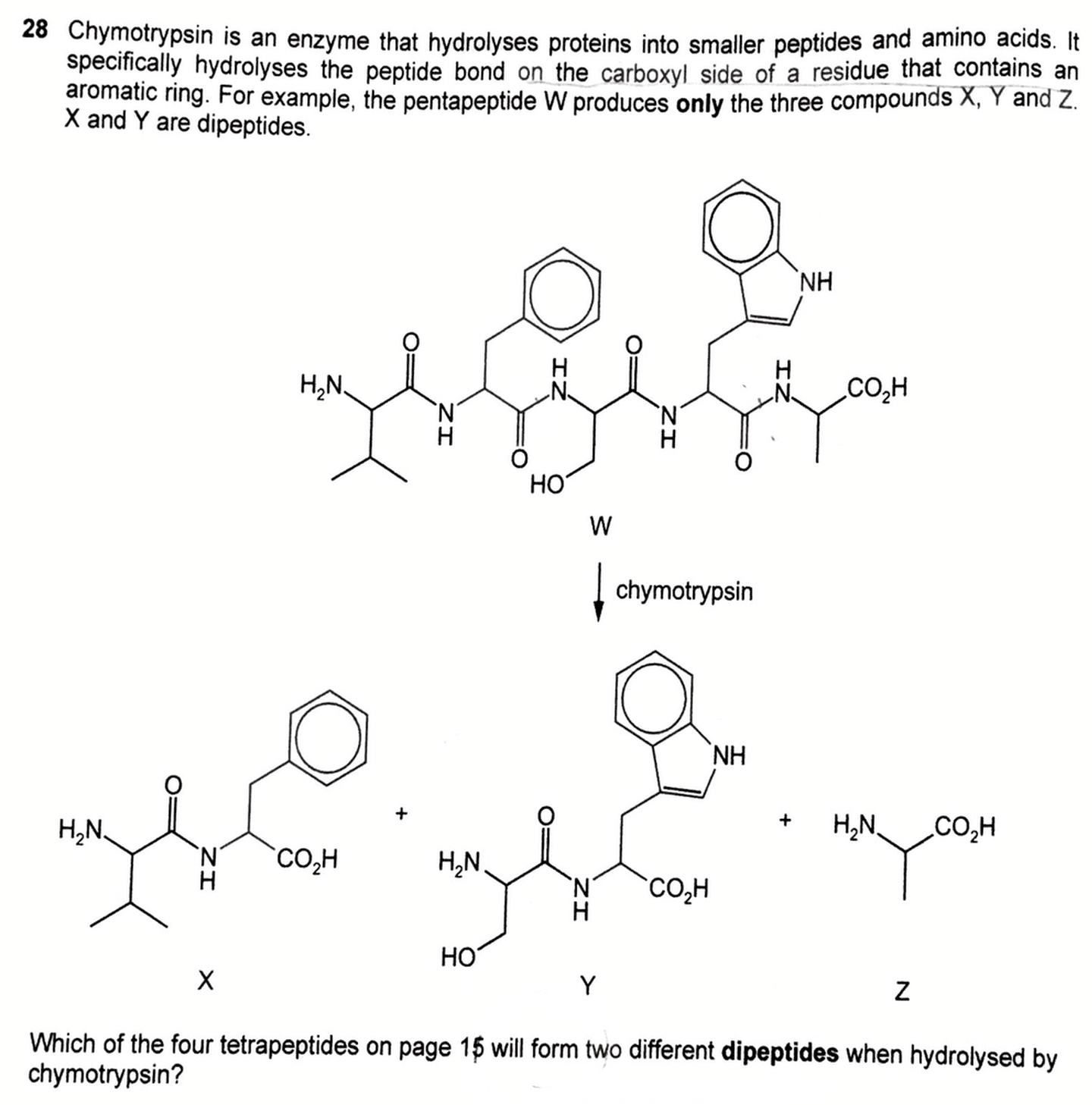
Given the activity of chymotrypsin, we need to deduce which option will form 2 dipeptides when hydrolysed by the enzyme.
First let's interprete the struture of pentapeptide W.
Recall the primary structure of proteins has a very predictable repeat unit:
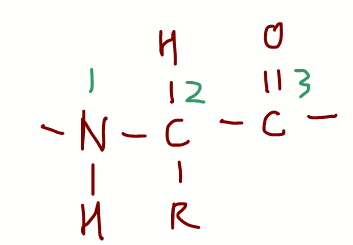
Position 1 - amine group
Position 2 - carbon with R group
Position 3 - acid group
Therefore we can find out the positions 1, 2 and 3 for W.

Next, we know that chymotrypsin will hydrolyse the peptide bond on the acid or carboxyl side of a residue with an aromatic ring.
So we can look at W, find the R groups with aromatic ring, break the peptide bond on the acid side, and figure out the products X (dipeptide), Y (dipeptide) and Z (amino acid) formed.

Once we understand how chymotrypsin functions, we can now apply this to the options and see which one gives us 2 dipeptides when hydrolysed with chymotrypsin.
A will give 1 tripeptide and 1 amino acid, hence not the answer.

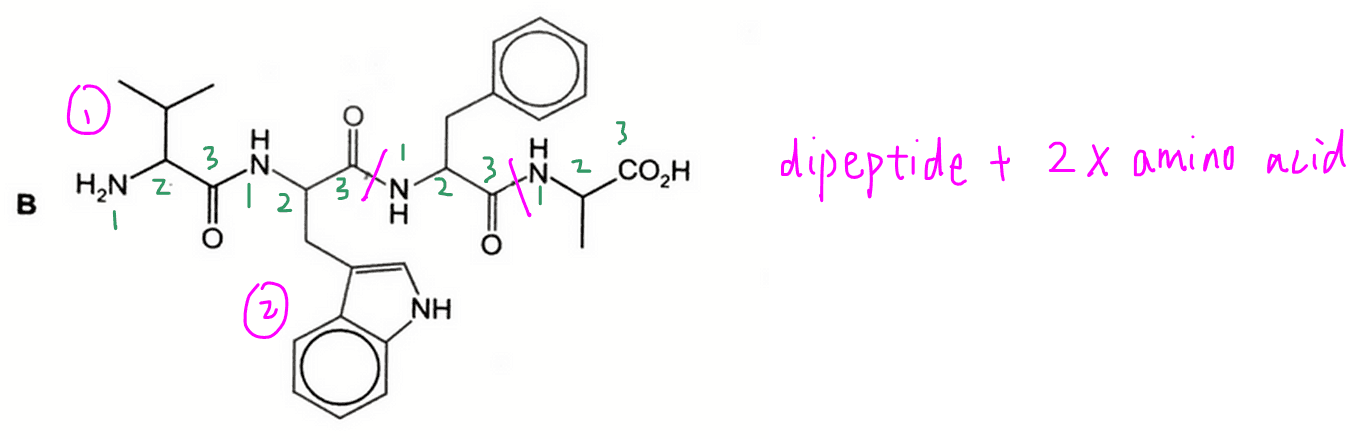
B will give 1 dipeptide and 2 amino acids, hence not the answer.
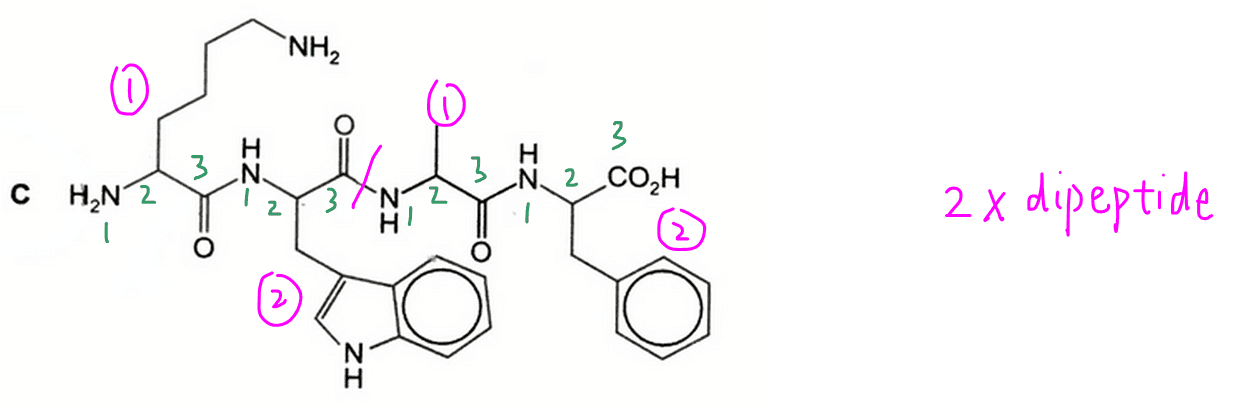
C will give 2 dipeptides hence this is the answer that we want.
D will give 2 amino acids and 1 dipeptide, hence not the answer.

Topic: Nitrogen Compounds and Proteins, Organic Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2019 A Level H2 Chemistry Paper 1
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's top H2 Chemistry tuition choice since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or online tuition classes!
