Deduce Major Product for Electrophilic Addition of Propene
Let Chemistry Guru, Singapore's esteemed A Level Chemistry tuition centre, guide you through our discussion question this week!
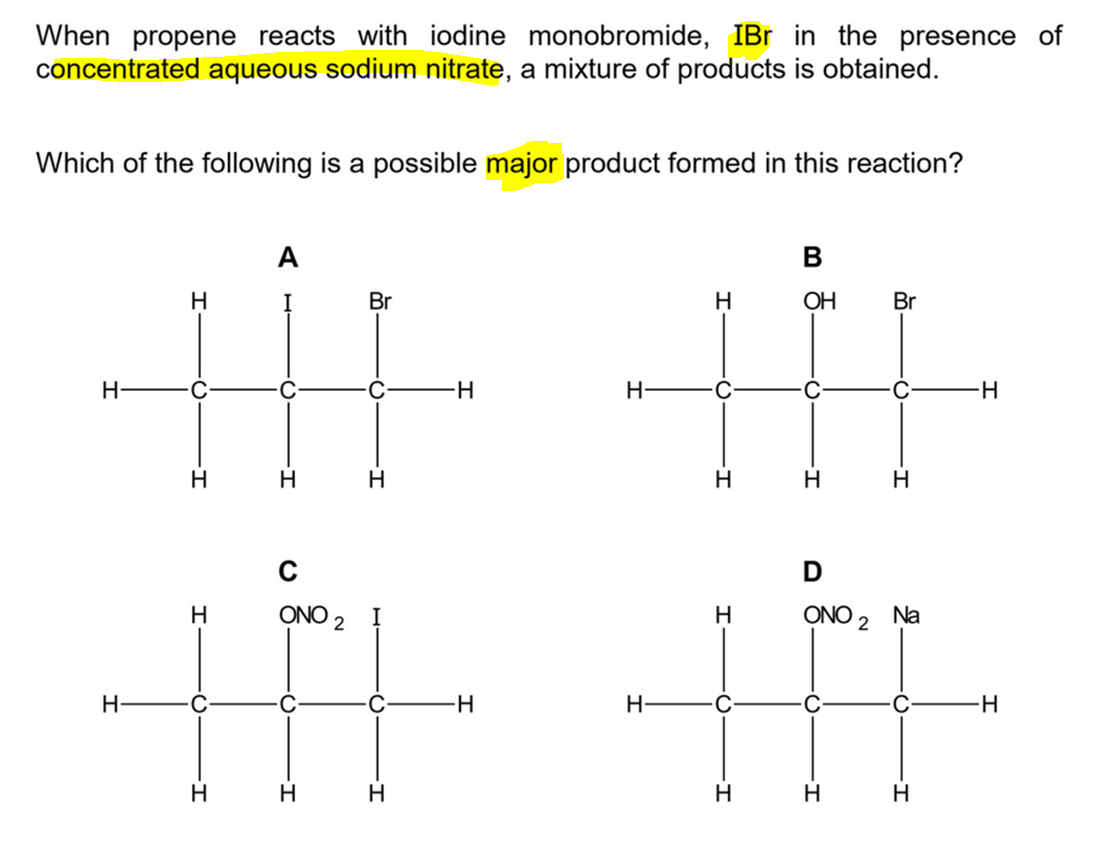
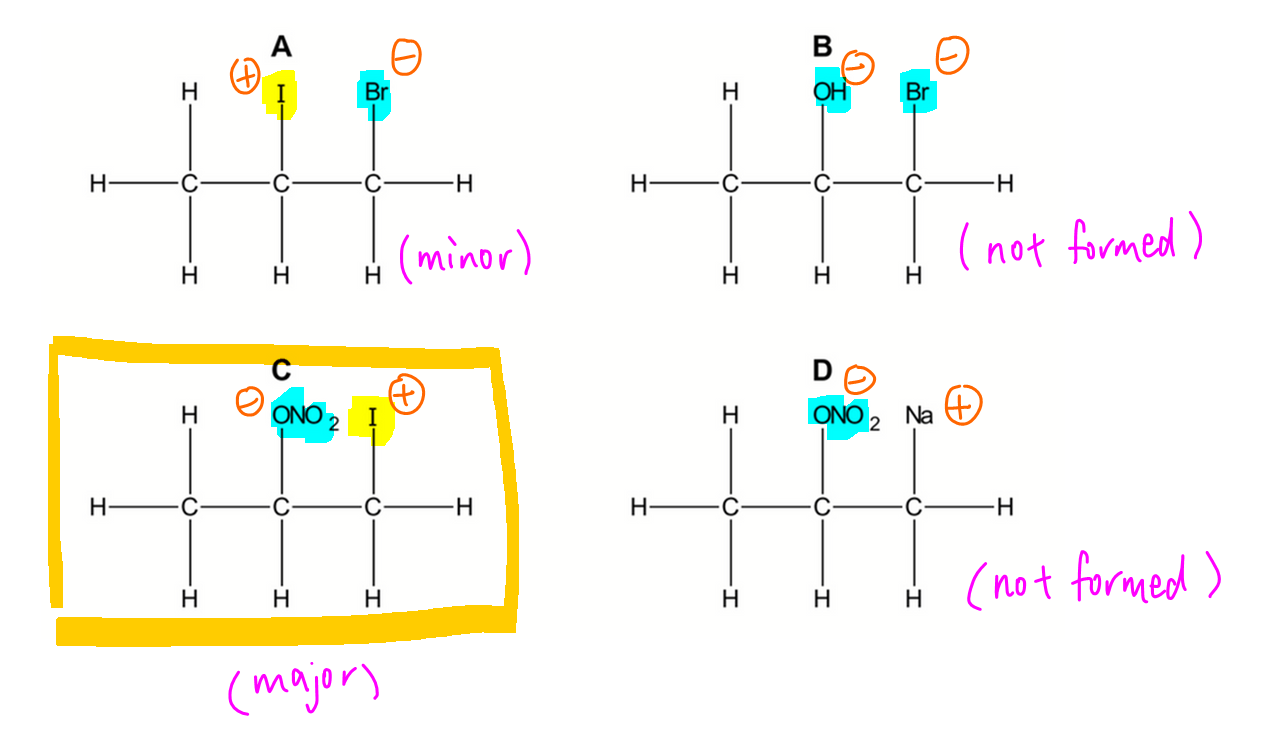
We want to determine the major product formed when propene reacts with iodine monobromide in concentrated aqueous sodium nitrate.
Notice this reagents and conditions is not in syllabus and we do not need to memorise it.
Therefore we have to apply our understanding of electrophilic addition of alkene to deduce the products formed.
Check out this video lesson to learn electrophilic addition of alkenes in detail.:
1. Deduce electrophile in Step 1
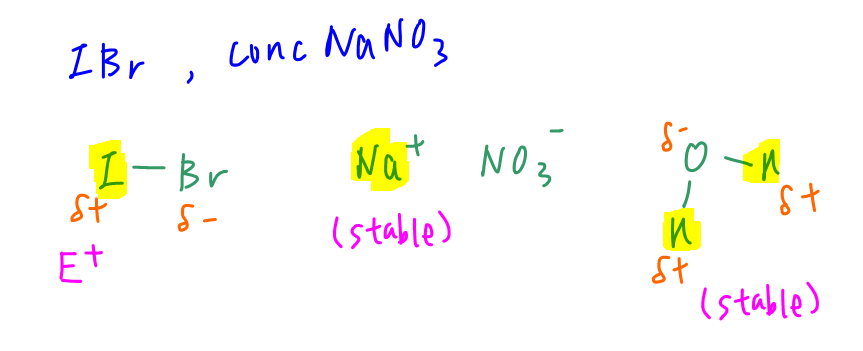
Only iodine in IBr is partially positive charged and reactive enough to act as electrophile.
Na+ is very stable and hydrogen in water is relatively stable so we will ignore Na and H.
Therefore the first step of electrophilic addition will be:
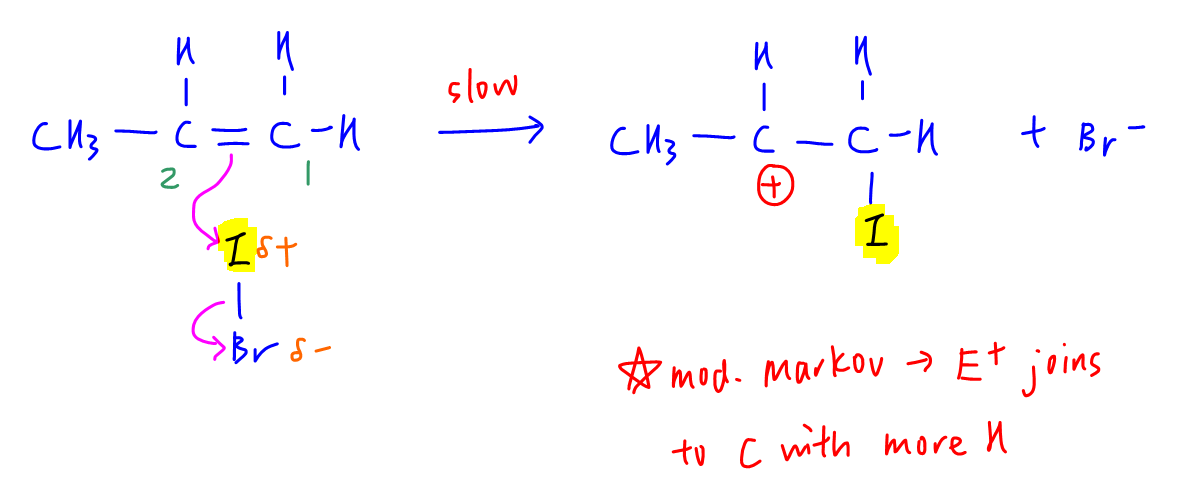
Electrophile iodine will join to carbon C1 with more hydrogen to form a more stable carbocation.
We can deduce this via modified Markovnikov Rule where electrophile joins to the carbon with more hydrogen during electrophilic addition of alkene to give the major product.
2. Deduce nucleophile in Step 2
Now the negative species Br- (from IBr), NO3- (from conc NaNO3) and OH- (from water) can attack the carbocation to form different products.
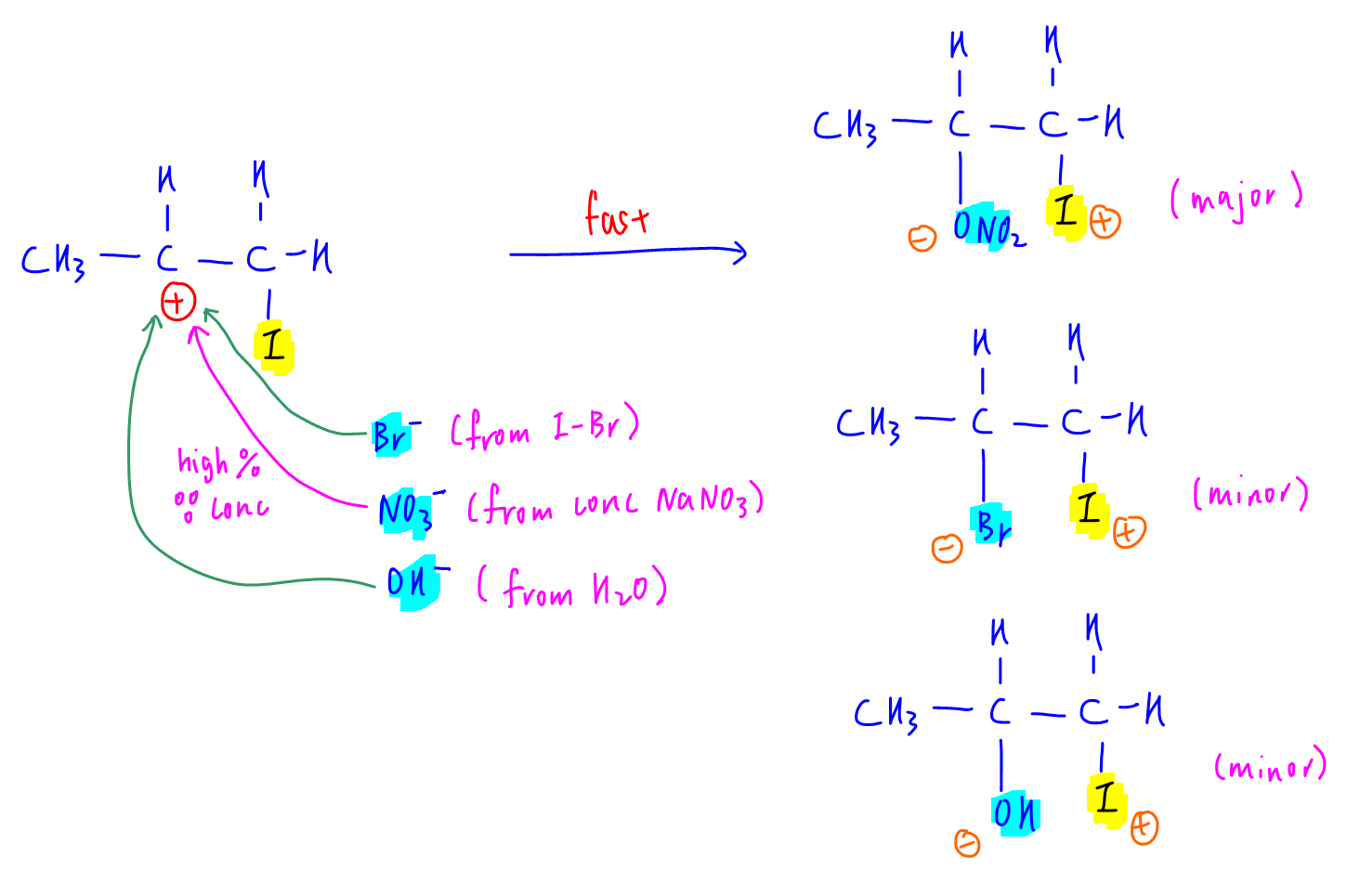
Since NO3- is concentrated, the probability of it attacking the carbocation is the highest to give the major product.
Therefore the major product formed will be option C.
Topic: Alkenes, Organic Chemistry, A Level Chemistry, Singapore
Back to other previous Organic Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's top JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
