Deduce Propagation Step for Free Radical Substitution of Ethane
Let Chemistry Guru, Singapore's esteemed A Level Chemistry tuition centre, guide you through our discussion question this week!
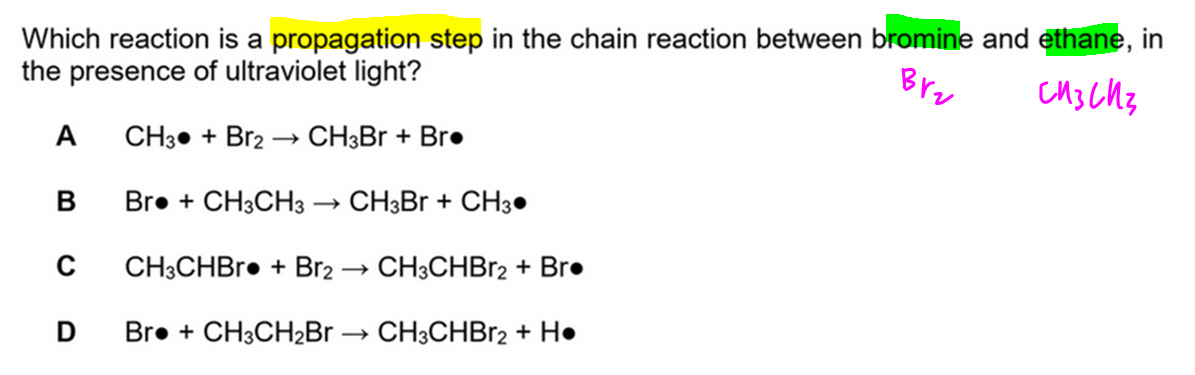
We want to deduce which is a propagation step in the free radical substitution between bromine and ethane.
Check out the following video to learn more about FRS mechanism of alkane.
Let's recap the features of propagation step.
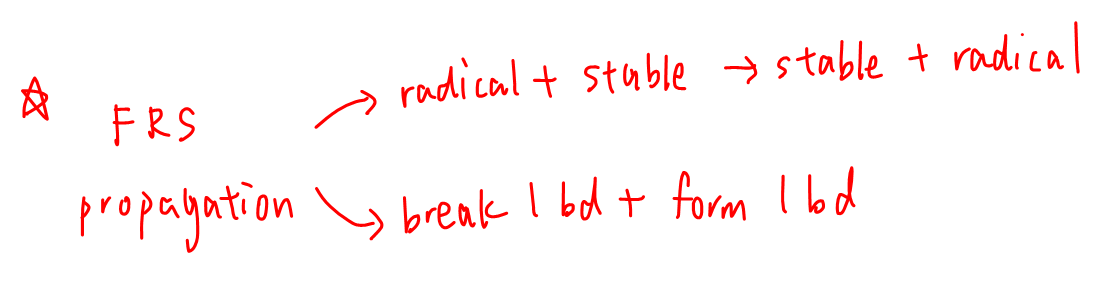
1. The number of reactive radicals stay the same hence the reaction continues or propagates.
2. Only involves breaking 1 bond and forming 1 bond homolytically.
Here are the standard propagation steps for monosubstitution of ethane.
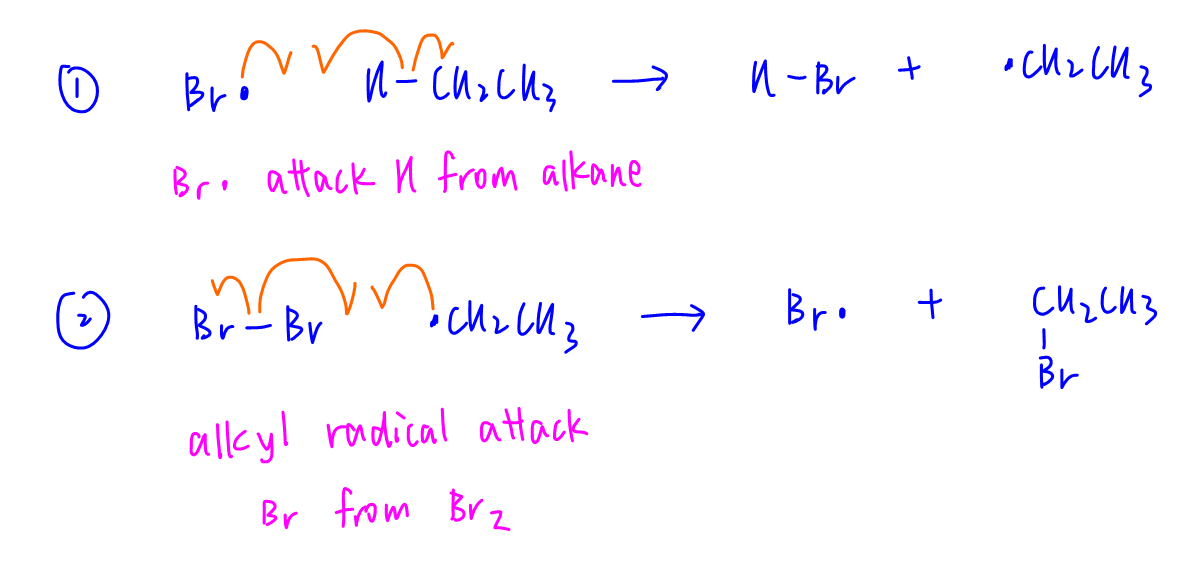
1. Bromine radical attacks ethane and takes H to form HBr and ethyl radical
2. Ethyl radical attacks bromine molecule and takes Br to form bromoethane and bromine radical
The bromine radical is regenerated to attack another ethane for another monosubstitution, or bromoethane for disubstitution.
Let's run through the options and see which is a possible propagation step.
A. CH3• + Br2 → CH3Br + Br•
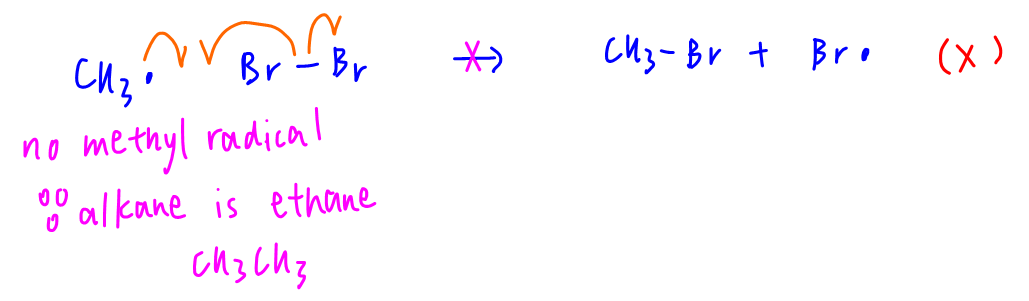
Since we are using ethane, there is no methyl radical formed during the reaction.
Hence option A is wrong.
B. Br• + CH3CH3 → CH3Br + CH3•

This involves breaking C-C bond which is not observed during FRS.
Hence option B is wrong.
C. CH3CHBr• + Br2 → CH3CHBr2 + Br•
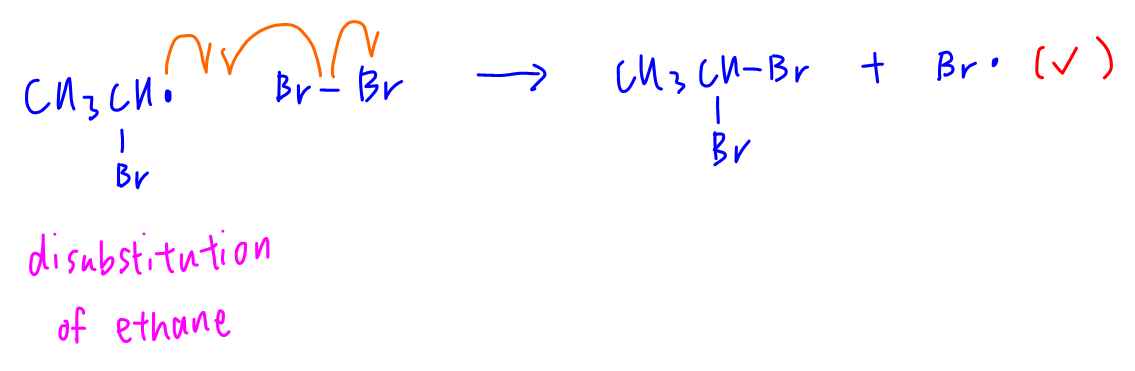
This step is possible during disubstitution of ethane from CH3CH2Br to CH3CHBr2.
Hence option C is correct.
D. Br• + CH3CH2Br → CH3CHBr2 + H•

When bromine radical attacks alkane, it should take the hydrogen since hydrogen is more exposed.
Hence we should form HBr and alkyl radical as products instead.
In FRS, H radicals are not formed hence option D is wrong.
Therefore the answer to this question is option C.
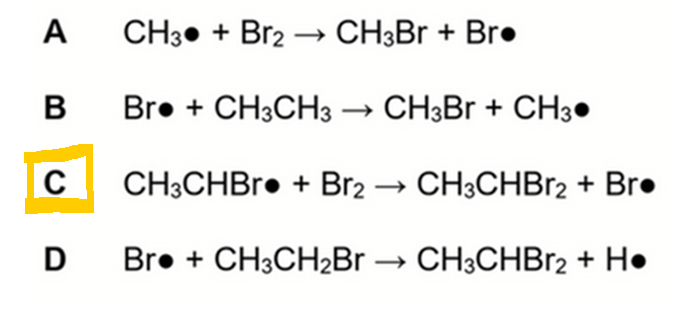
Topic: Alkanes, Organic Chemistry, A Level Chemistry, Singapore
Back to other previous Organic Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's top JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
