Deduce Ratio of Monosubstituted Products for Free Radical Substitution
In this JC1 webinar we want to learn how to deduce the number of monosubstituted products for FRS and the ratio of these products.
Let Chemistry Guru, Singapore's renowned JC Chemistry tuition centre, guide you through our example propane, C3H8.
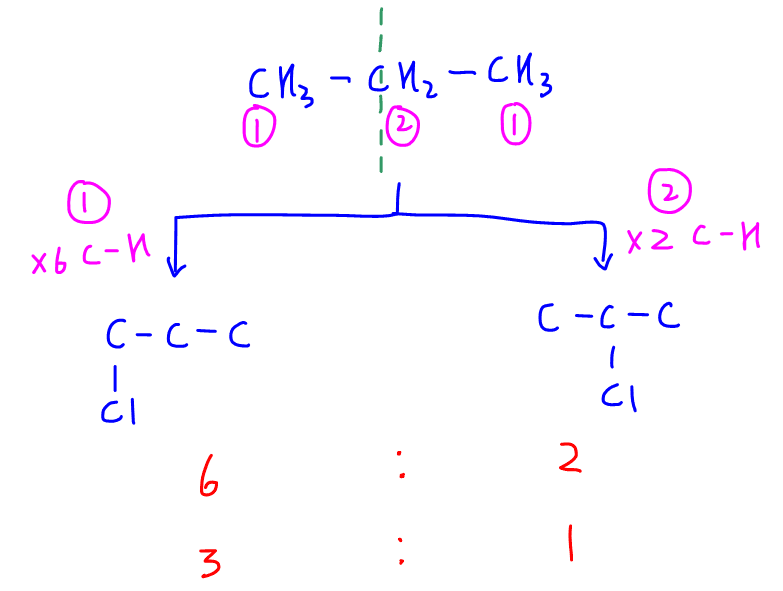
Notice there are 2 different types of carbons in propane which we have labelled as carbon-1 and carbon-2.
Assuming the probability of substitution at each C-H bond is equal:
- substituting 6 of these hydrogens at both of carbon-1 will give 1-chloropropane
- substituting 2 of these hydrogens at carbon-2 will give 2-chloropropane
Hence the ratio of 1-chloropropane to 2-chloropropane will be 6:2 or 3:1.
Sometimes the reactivity of different types of carbons are given, so we have to take that into consideration as well.
In general the reactivity increases from primary carbon to secondary carbon to tertiary carbon.
Let's say for example the relative reactivity is:
1 for primary carbon
4 for secondary carbon
7 for tertiary carbon
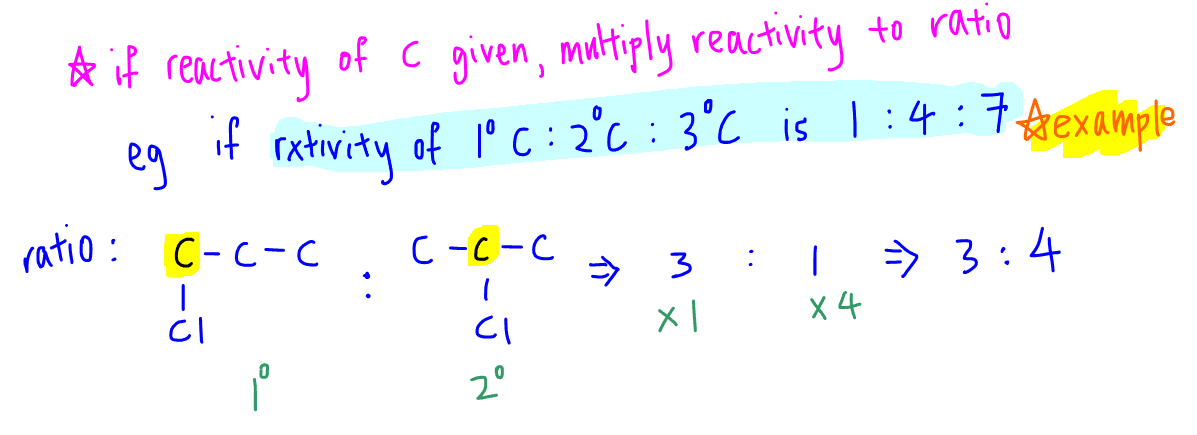
We then need to determine the type of carbon and multiply its probability by its reactivity.
Carbon-1 that forms 1-chloropropane is a primary carbon, so we multiply its probability 3 by reactivity 1 to give final proportion 3.
Carbon-2 that forms 2-chloropropane is a secondary carbon, so we multiply its probability 1 by reactivity 4 to give final proportion 4.
Finally the ratio of 1-chloropropane to 2-chloropropane, after taking into consideration of reactivity, will be 3:4.
Topic: Alkanes, Organic Chemistry, A Level Chemistry, Singapore
Back to other previous Organic Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's trusted JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
