Determine pH of AgOH from Solubility Product
Let Chemistry Guru, Singapore's top choice for JC Chemistry tuition, guide you through this week's question.
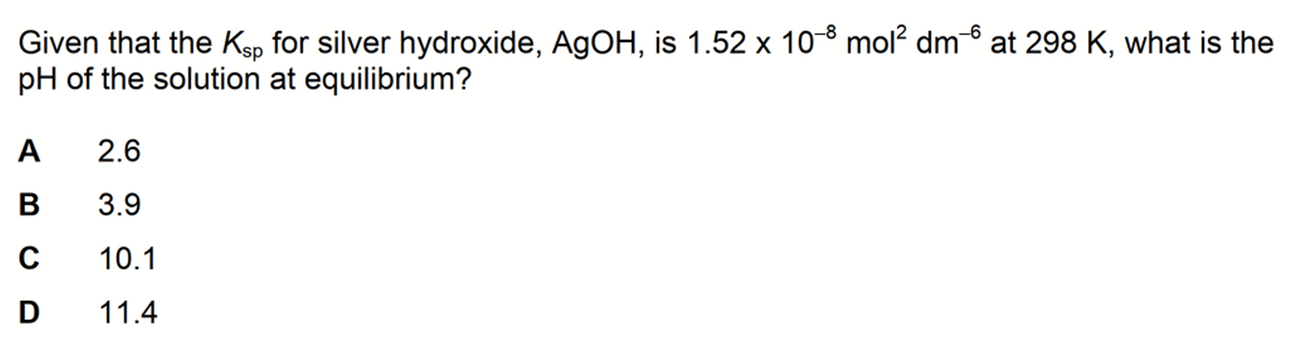
We are required to determine the pH of silver hydroxide solution at equilibrium.
Since silver hydroxide is alkaline, we can rule out options A and B straight away.
Let's consider the relationship between solubility product Ksp and solubility.
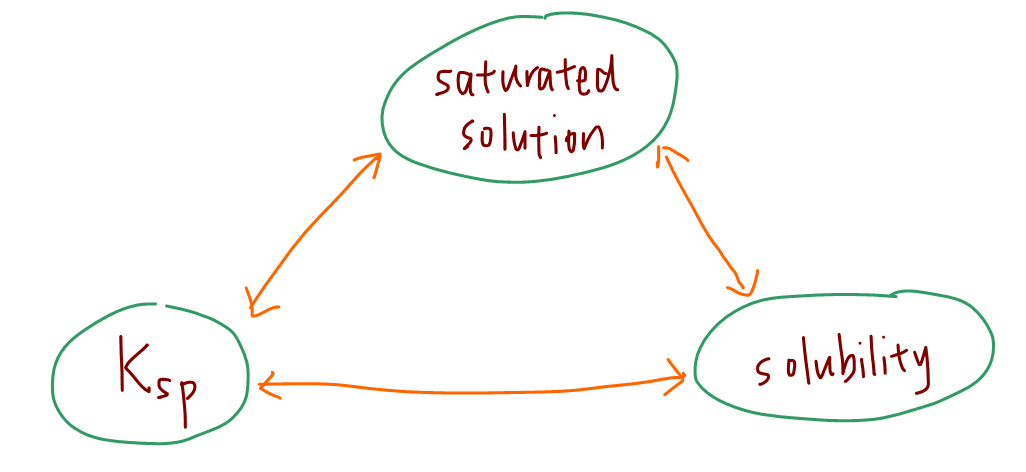
When the solution is saturated, concentration of ions in solution is at maximum and constant.
Hence a saturated solution is at equilibrium and it's meaningful to discuss Ksp which is an equilibrium constant.
A saturated solution is also related to its solubility or maximum amount of salt that can dissolve per unit volume of solvent.
Hence Ksp and solubility are related to each other.
Check out the following video lesson for a more in-depth discussion involving the relationship between solubility product and solubility.
We can write out the dissociation of AgOH and determine the concentration of its ions in terms of solubility x.

Then we can write out Ksp expression and determine solubility x in terms of Ksp.
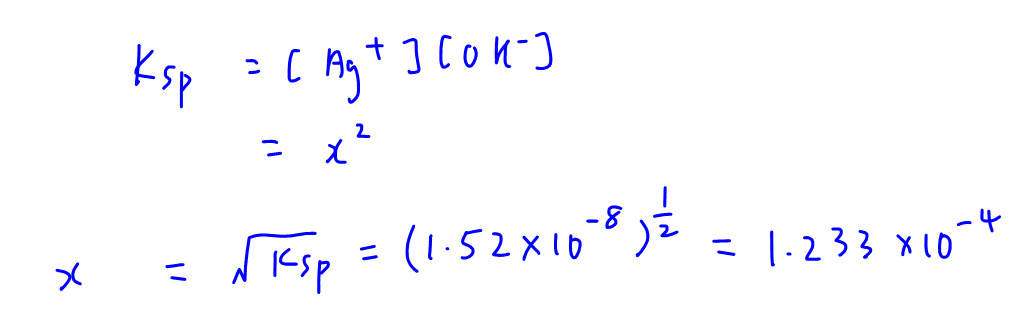
OH- concentration has the same value 1.233 x 10-4 and hence we can calculate pOH and pH of the solution.

Therefore the answer to this question will be option C.
Topic: Solubility Product, Physical Chemistry, A Level Chemistry, Singapore
Back to other previous Physical Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's esteemed H2 Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
