Determine Equilibrium Constant Using ICE Table Method
In this video created by Chemistry Guru, Singapore's leading JC Chemistry tuition centre, let's go through how to determine equilibrium constant Kc using the ICE table method.
Given this simple example:
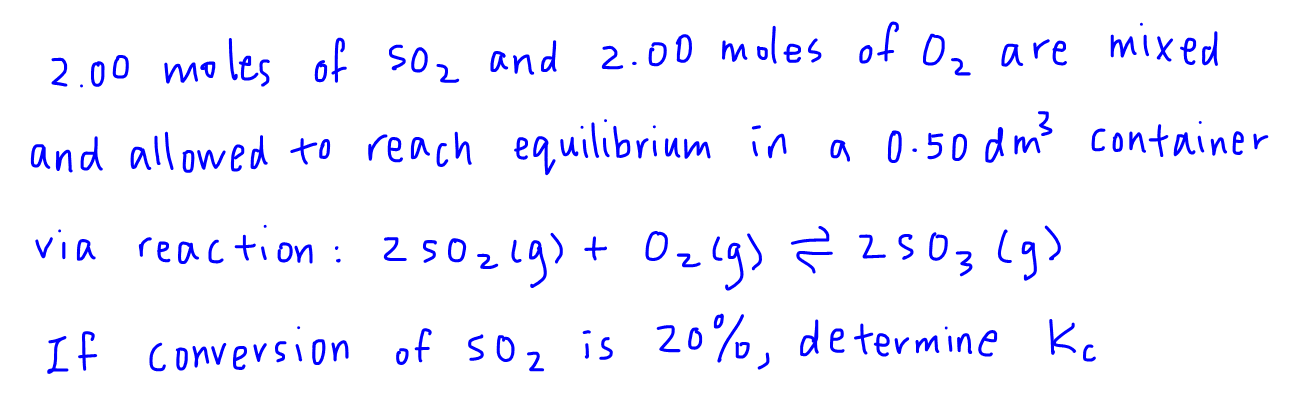
First we have to write out the balanced equation and 3 additional rows which represents the following:
I - INITIAL number of moles
C - CHANGE in number of moles
E - EQUILIBRIUM number of moles
The initial moles of SO2 and O2 are both given as 2.00 moles, so we call put that info into the ICE table.
Also the conversion of SO2 is 20% which works out to be 0.4 moles of SO2 reacted, and we can put that in as well.
So this will be what the ICE table would look like at this stage:
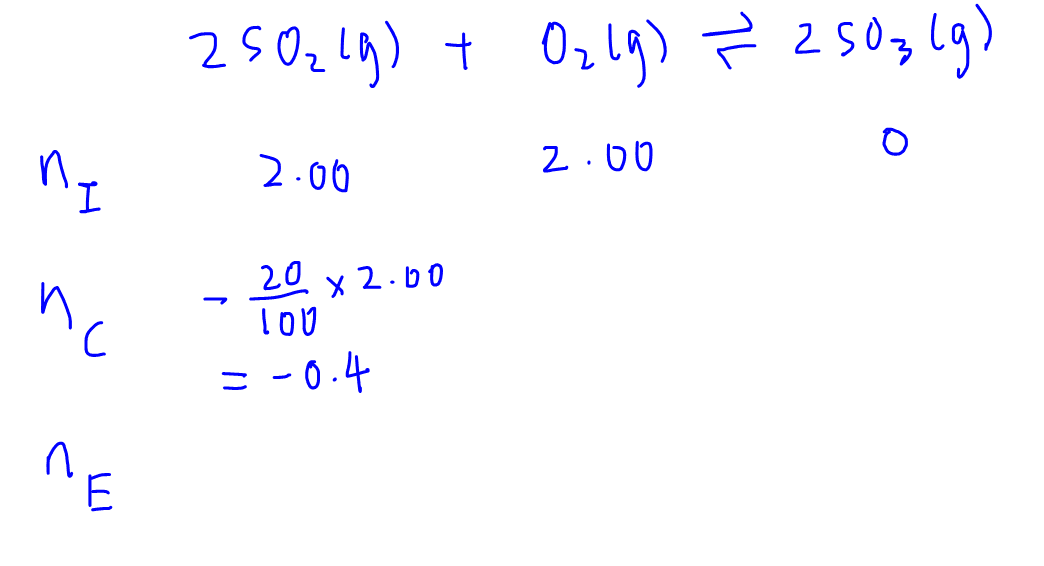
Once we have the change in moles of SO2, we can determine the change in moles for the rest of the reactants and products.
Since the mole ratio for all species in the balanced equation is
2:1:2 (2SO2 reacted; to 1O2 reacted; to 2SO3 formed),
the mole ratio for the CHANGE row will also follow the same mole ratio
2:1:2 (0.4 moles of SO2 reacted; to 0.2 moles of O2 reacted; to 0.4 moles of SO3 formed)
So we can fill up the CHANGE row and then determine the last row for equilibrium moles of all reactants and products.
We can then divide the EQUILIBRIUM row by the volume of container to determine the concentration of all species at equilibrium.
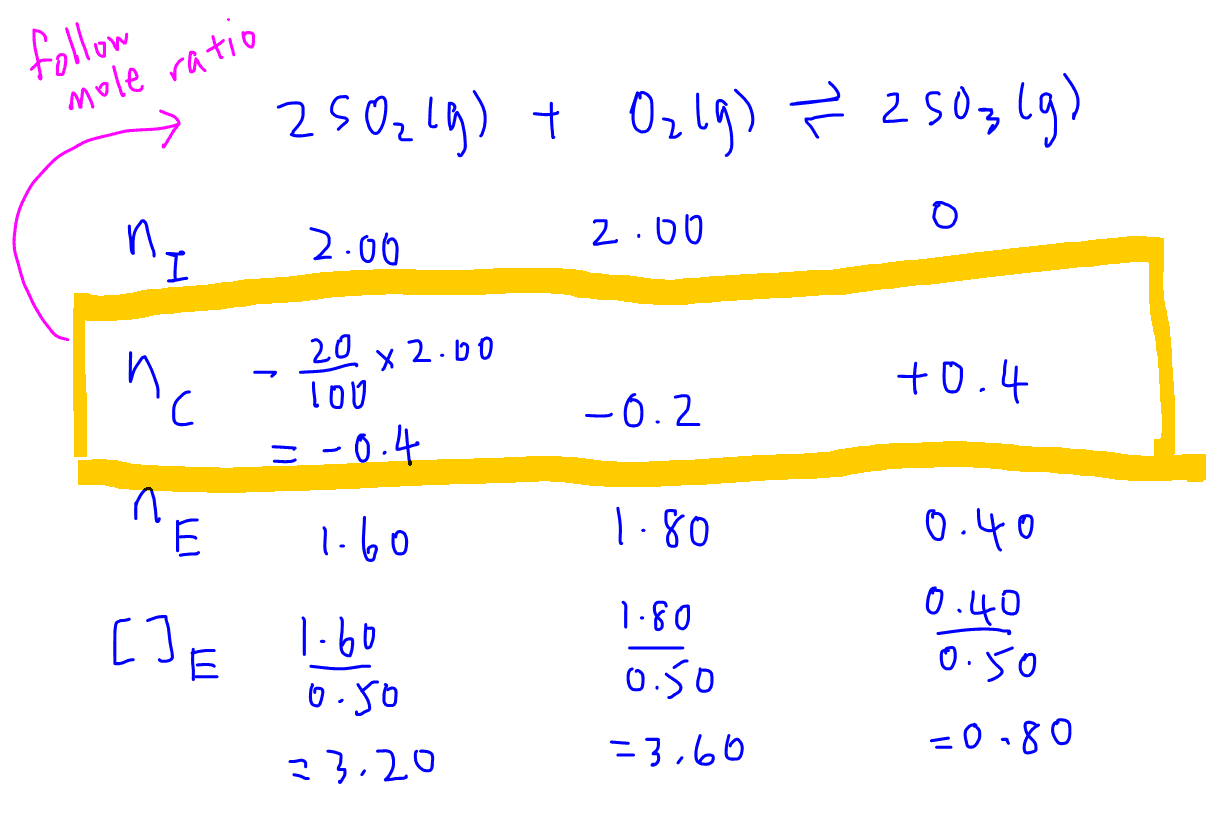
Finally we can write out the Kc expression and substitute in all the concentration values to calculate the equilibrium constant for this exercise.
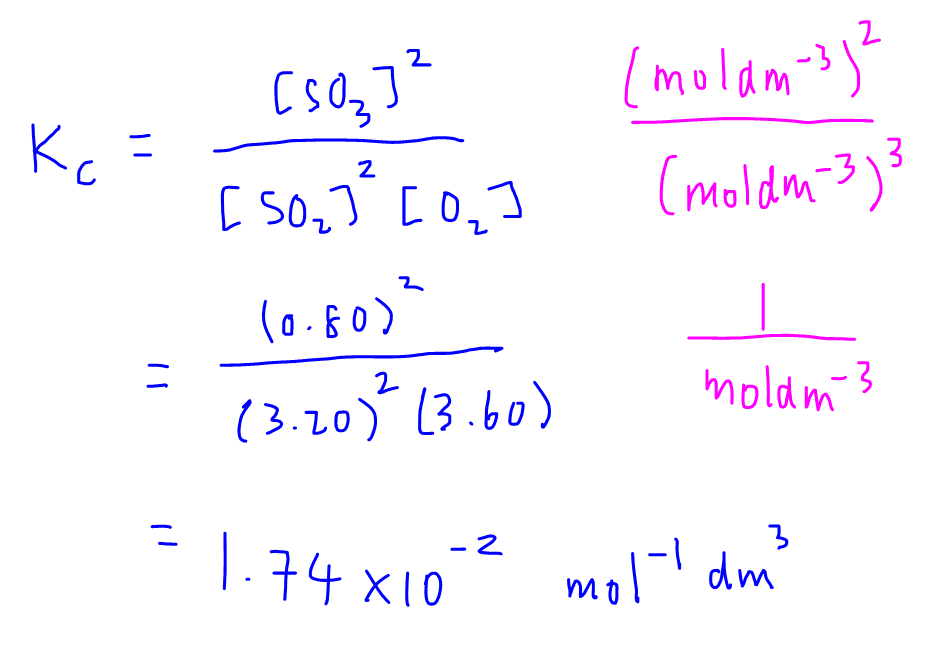
Topic: Chemical Equilibria, Physical Chemistry, A Level Chemistry, Singapore
Back to other previous Physical Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's top H2 Chemistry tuition choice since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my A Level H2 Chemistry Tuition classes at Bishan or online tuition classes!
