Determine Limiting Reagent
In this video we want to discuss how to determine the limiting reagent for mole concept questions, and use the limiting reagent to determine the amount of products formed.
Let Chemistry Guru, Singapore's top JC Chemistry tuition centre, guide you through an example:

It is important for students not to assume that all the Na2CO3 will completely react with all the HCl.
We have to determine if one of the reactants is limiting, and therefore the amount of CO2 formed is based on the limiting reagent.
First let's calculate the amount of both reactants.
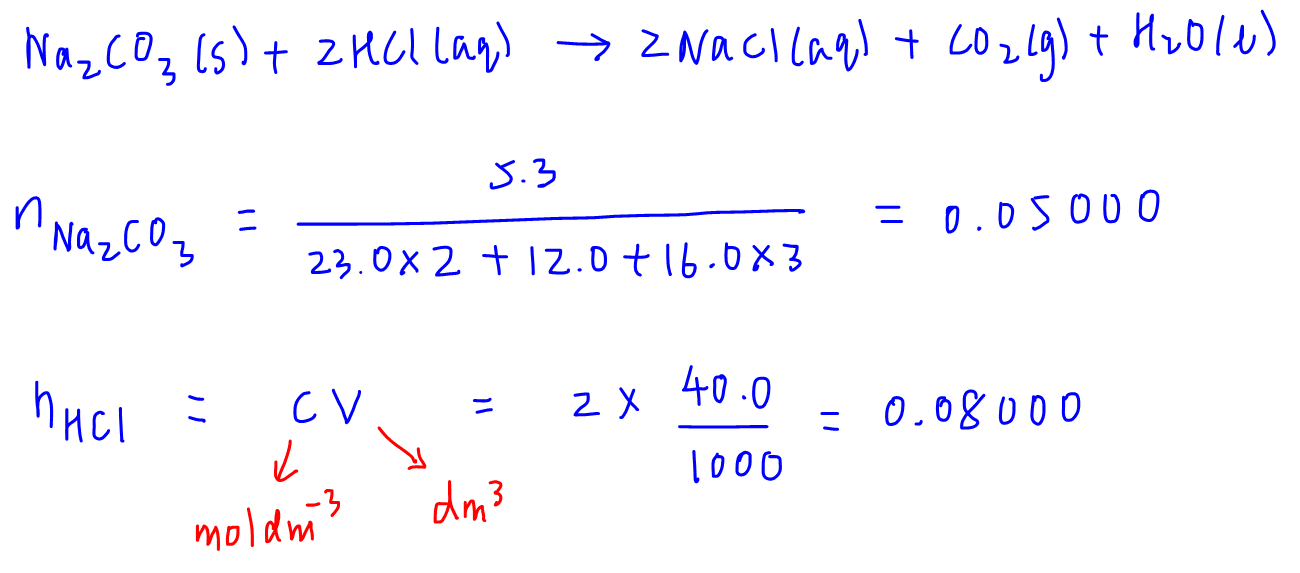
Next, to determine the limiting reagent, we calculate the amount of reactant B required to completely react with all of reactant A.
In this case if we react 100% of Na2CO3 which is 0.05000 mol, how much of HCl is required?
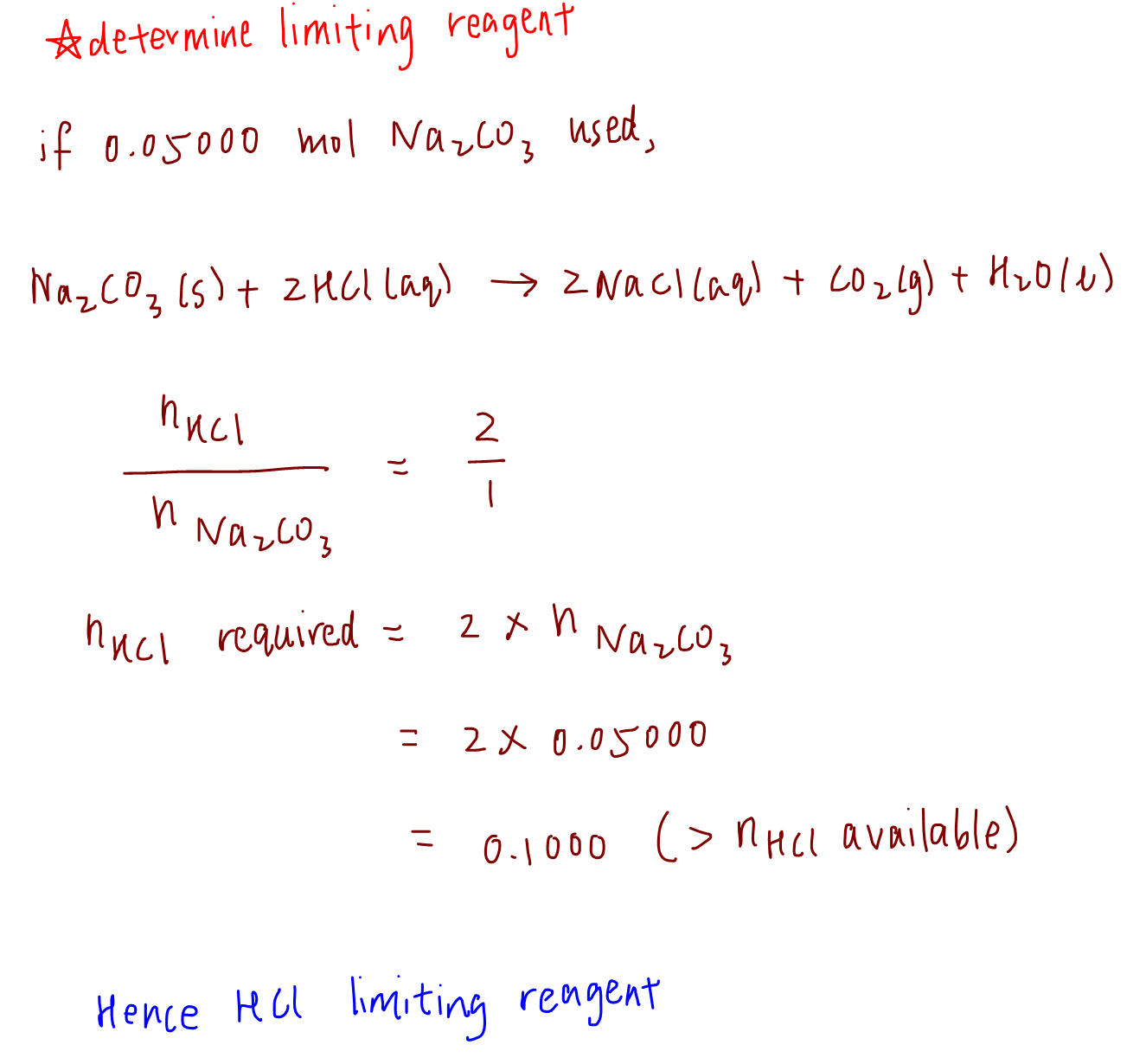
Based on the calculation, we need 0.1000 mol of HCl to react with 100% of Na2CO3, but we only have 0.08000 mol of HCl.
This means that HCl is limiting and Na2CO3 is in excess.
Sidenote:
If based on the calculation we have enough HCl to react with 100% of Na2CO3, this means after all the Na2CO3 is used up, we will still have excess HCl left.
In this case then Na2CO3 is limiting and HCl is in excess.
Finally we can use the limiting reagent to determine the amount and volume of CO2 produced.

For the detailed step-by-step discussion on how to determine limiting reagent, check out this video!
Topic: Mole Concept, Physical Chemistry, A Level Chemistry, Singapore
Back to other previous Physical Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's reputable A Level Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my A Level H2 Chemistry Tuition classes at Bishan or online chemistry classes!
