Determine Mass of Black Powder Needed to Fire Cannonball
Let Chemistry Guru, Singapore's top choice for JC Chemistry tuition, guide you through this mole concept question.

We are required to calculate the mass of black powder needed to expel the cannonball.
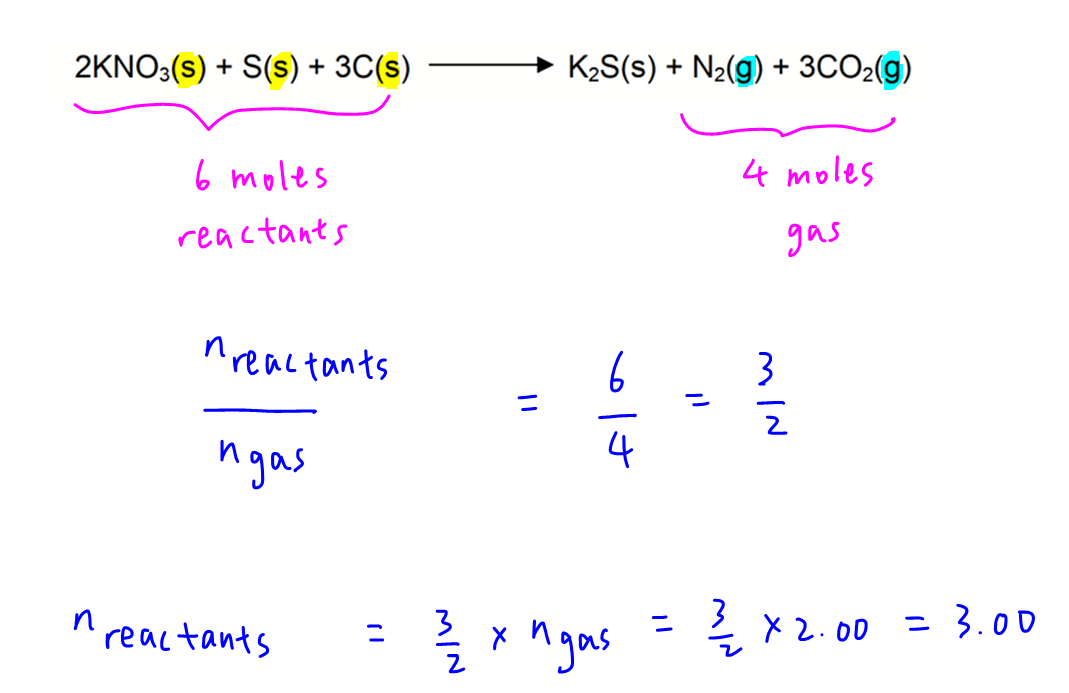
6 moles of reactants will give 4 moles of gaseous products.
Hence by comparing mole ratio, we can calculate moles of reactants to be 3.00 mol.
Next we need to split these 3.00 mol of reactants to the individual parts.
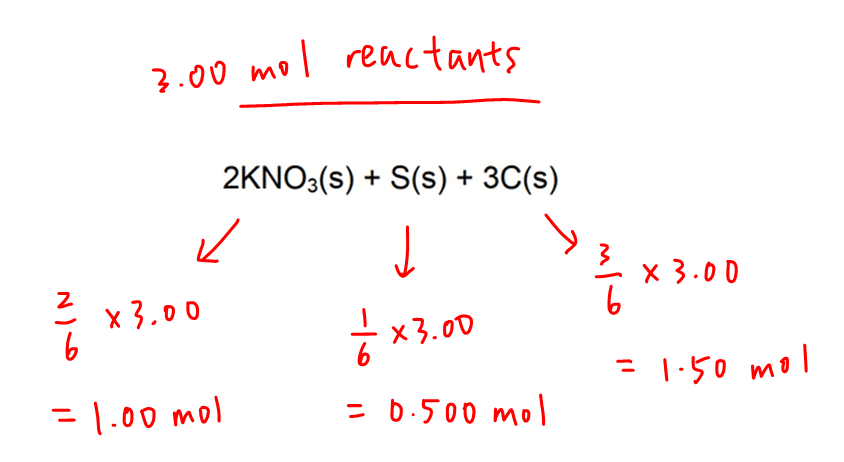
Out of 6 parts in total, there are:
- 2 parts KNO3 hence moles of KNO3 is 1.00 mol
- 1 part S hence moles of S is 0.500 mol
- 3 parts C hence moles of C is 1.50 mol
We can then find the mass of each reactant and combine them to find the mass of black powder required to be 135.2g.

Hence the answer to this question will be option B.

There is an alternative way to compare the mole ratio.
We can treat 2 moles of KNO3, 1 mole of S and 3 moles of C to be one unit of black powder.
This means the mole ratio of black powder to gaseous products is now 1 to 4.
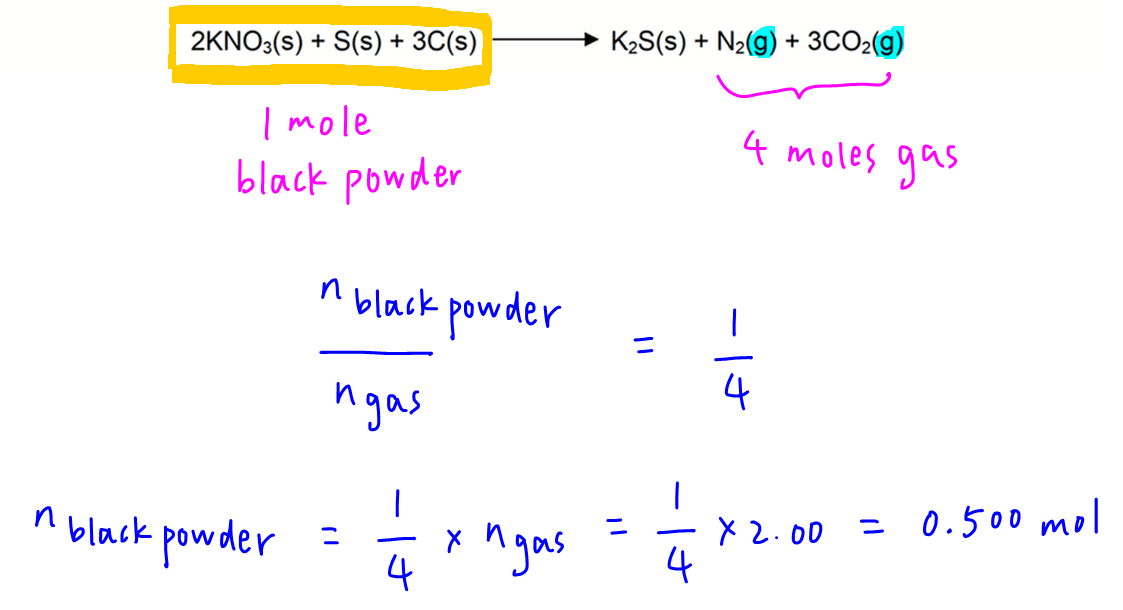
Therefore moles of black powder will be 0.500 mol.
We can then find the molar mass and hence the mass of black powder which will be the same value at 135.2g.

Topic: Mole Concept, Physical Chemistry, A Level Chemistry, Singapore
Back to other previous Physical Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's esteemed H2 Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
