2019 A Level H2 Chemistry Paper 1 Question 7 - Determine Gas Volume Using Ideal Gas Equation
Here's question 7 from 2019 A Level H2 Chemistry Paper 1:

We are required to determine the volume of nitrogen gas produced by the decomposition of sodium azide in airbags.
Given that the products are sodium metal and nitrogen gas, we can work out the balanced equation for this decomposition.

We can find the number of moles of sodium azide from its mass, which is 0.07692 mol.
Comparing mole ratio of nitrogen gas to sodium azide, we can calculate the amount of nitrogen gas to be 0.1153 mol.
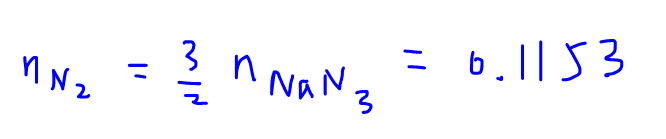
We can then make use of Ideal Gas Equation to determine volume of nitrogen gas.
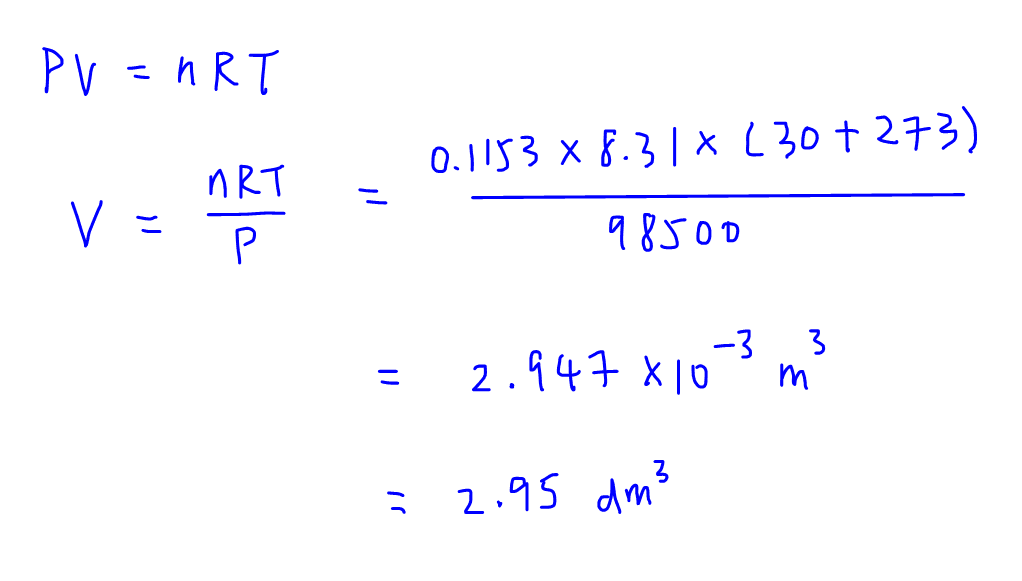
Take note all the terms have to be in SI units:
P is pressure in Pa
V is volume in m3
n is number of moles
R is gas constant 8.31 J K-1 mol-1
T is temperature in K
Finally we have to convert volume from m3 to dm3 making use of the following conversion:
1 m3 = 1000 dm3
Therefore the answer to this question will be option D.
Topic: Gaseous State, Physical Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2019 A Level H2 Chemistry Paper 1
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's top H2 Chemistry tuition choice since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or online tuition classes!
