Drawing Dot and Cross Diagrams
In this video created by Chemistry Guru, Singapore's leading JC Chemistry tuition centre, we want to learn how to draw dot and cross diagrams for molecules and ions in Chemical Bonding.
Drawing dot-cross diagram is a fundamental and essential technique for us to determine the lewis structure and shape of molecules and polyvalent ions.
For A Level Chemistry Syllabus we now have elements that can expand octet, and species that can form dative bonds.
So we have to systematically deduce the arrangement of electrons instead of doing it by trial and error.
Guidelines for Drawing Dot Cross Diagram
There are a few guidelines that are useful:
1. The atom that needs the most electrons to be stable is the central atom.
2. For anions, electrons are added to the more electronegative atom. For cations, electrons are removed from the less electronegative atom.
3. The central atom will distribute its electrons to the surround atoms to make them stable or octet.
4. An atom will form the same number of bonds as the number of additional electrons required for stablility. For instance carbon needs 4 electrons to be octet so will form 4 bonds.
5. The central atom will expand octet to form more bonds if necessary. Remember more bonds means more energy is released so the species will be more stable.
6. Only Period 2 elements cannot expand octet, ie only these 4 elements carbon, nitrogen, oxygen and fluorine cannot expand octet. This means that octet rule is the exception and expansion of octet is the norm.

Here are some examples and their dot-cross diagrams and lewis structures.
1. NO3-
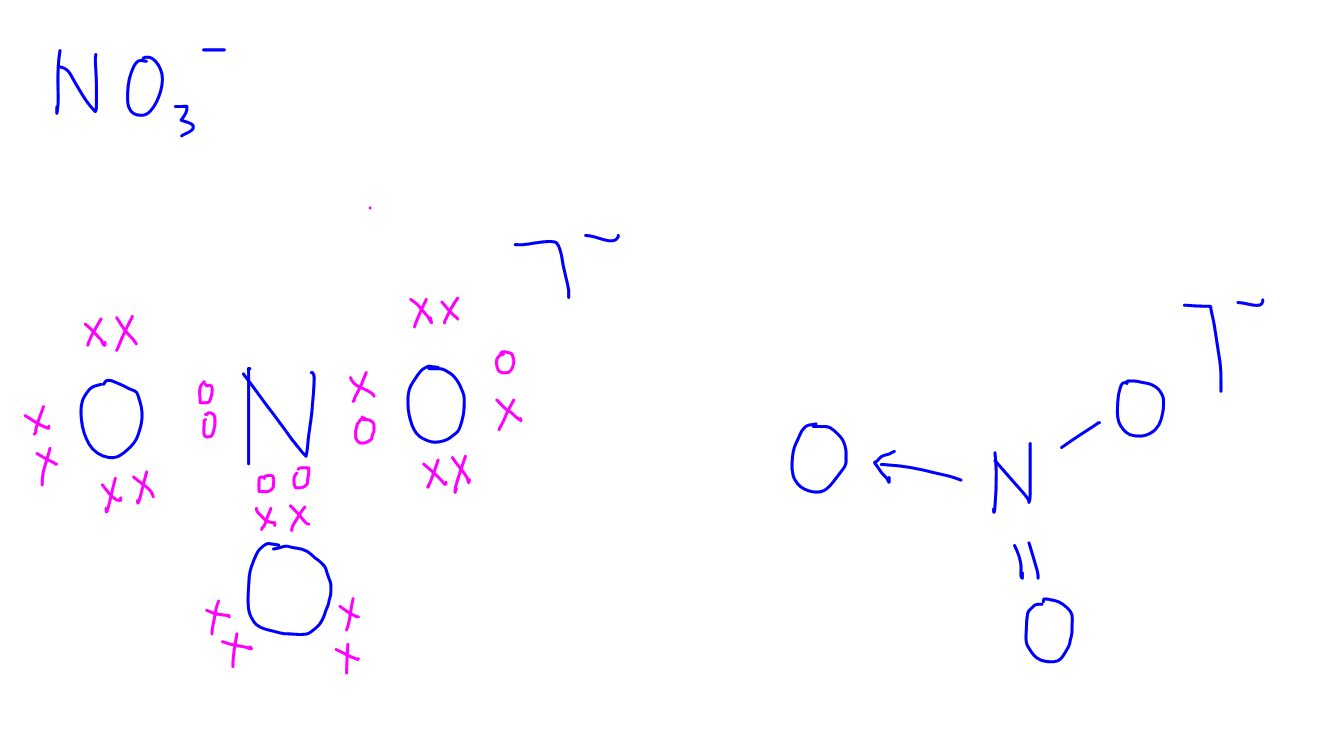
2. PCl4-
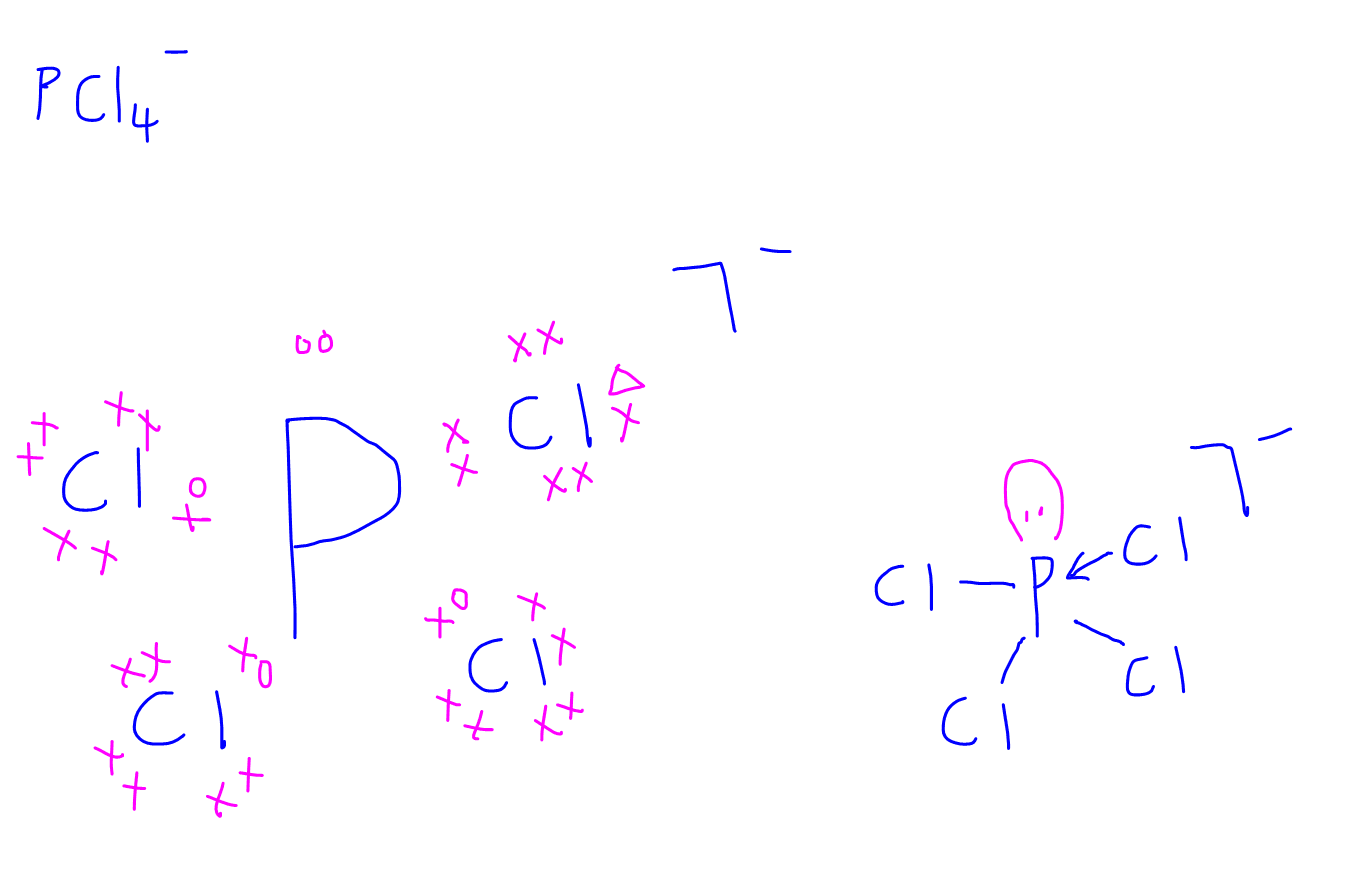
3. O3
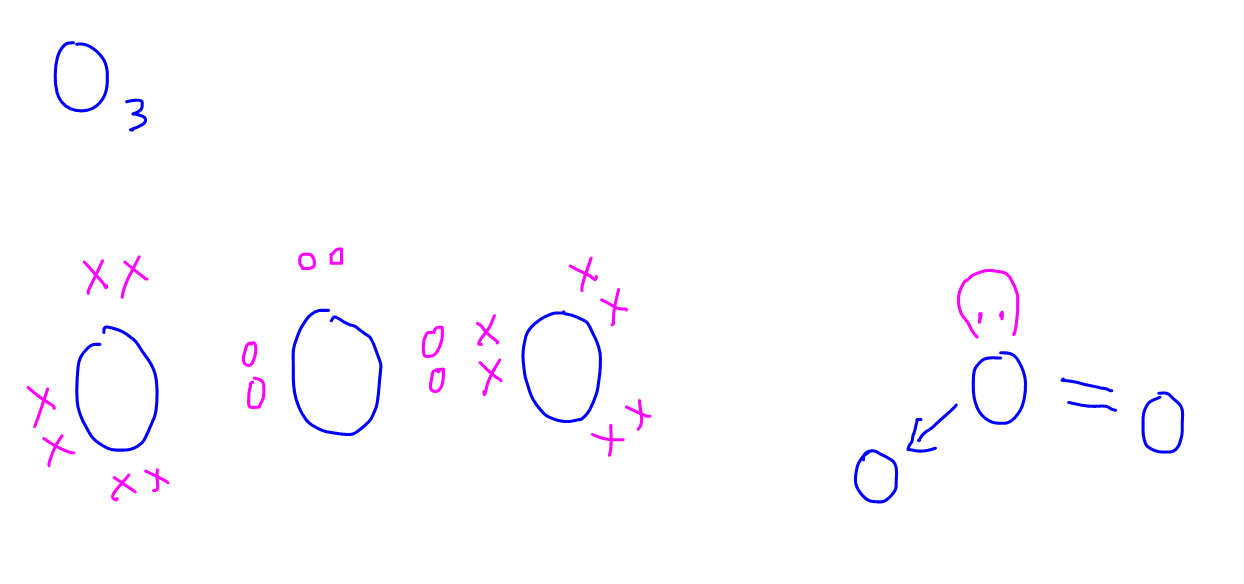
For the detailed step-by-step discussion on how to draw the dot and cross diagrams of these species, check out this video!
Topic: Chemical Bonding, Physical Chemistry, A Level Chemistry, Singapore
Back to other previous Physical Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's reputable A Level Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my A Level H2 Chemistry Tuition classes at Bishan or online chemistry classes!
