Alkene Mechanism: How to describe Electrophilic Addition Mechanism for Alkenes
One of the reaction mechanisms in A Level Chemistry Syllabus is Electrophilic Addition of Alkenes.
We need to be able to describe this mechanism in detail using curvy arrows.
Let's use the bromination of ethene as an example:

Electrophilic Addition Step 1 - Attack on electrophile to generate carbocation intermediate
Electron rich alkene will use the pi bond to attack the partially positively charged bromine atom which is closer to it.
A carbon-bromine bond is formed.
The other carbon which loses the electron will become positively charged, hence the name carbocation.
Bromide ion, Br- is also formed in the first step.
Take note that the first step is the slow step or rate determining step.

Electrophilic Addition Step 2 - Attack on carbocation by Br- to form product
The Br- formed will attack the carbocation and form the second carbon-bromine bond to form the product 1,2-dibromoethane.
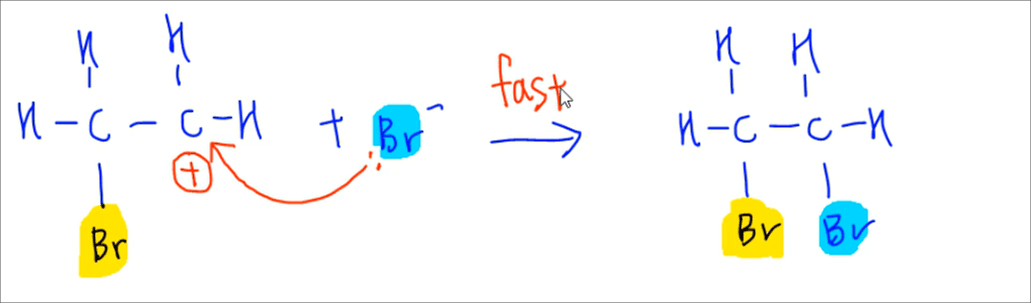
It's important to note that even though we are adding 2 bromine to alkene, they are added to the alkene differently:
a. First Br is added as an electrophile in the first step (highlighted yellow in the screenshot)
b. Second Br is added as a nucleophile in the second step (highlighted blue in the screenshot)
Check out this video lesson for a step-by-step tutorial to learn how to draw the mechanism via curly arrows!
Topic: Alkenes (Hydrocarbons), Organic Chemistry, JC, H2, A Level Chemistry, Singapore
Back to other previous Organic Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's leading JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or online chemistry classes!
