Electrophilic Addition with Aqueous Bromine
In this JC1 webinar we want to learn how to describe Electrophilic Addition mechanism when ethene reacts with aqueous bromine.
If you are not familiar with the mechanism of alkene do check out this video lesson on electrophilic addition.
There are 2 possible products, 2-bromoethanol (major) and 1,2-dibromoethane (minor).

First we have to deduce the electrophile since there are bromine and water present.
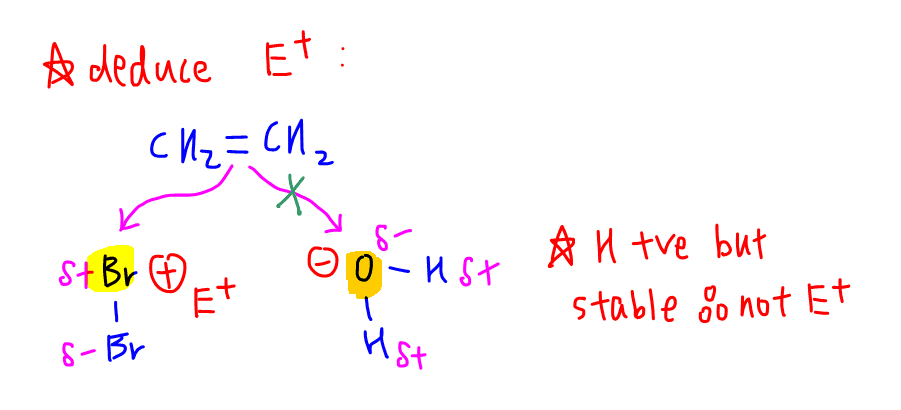
Bromine can act as electrophile since one of the bromine atoms can be partial positive charge.
Oxygen in water is more electronegative than hydrogen and will be partial negative charge, hence cannot be electrophile.
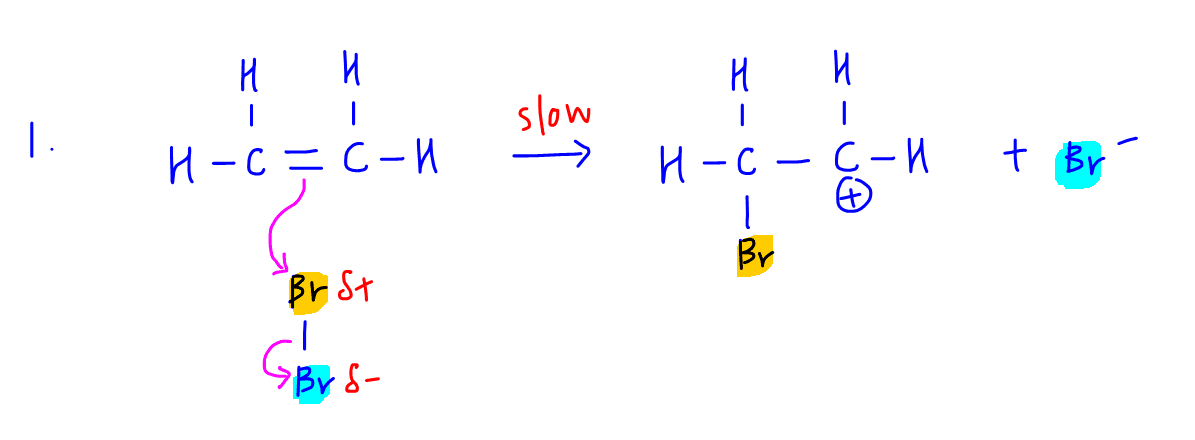
So we know step one will involve the ethene attacking electrophilic bromine to form carbocation intermediate and Br-.
Next we have to deduce the nucleophile that can attack the carbocation in step 2.
We have 2 possible nucleophiles - oxygen from H2O and Br- formed from step 1.
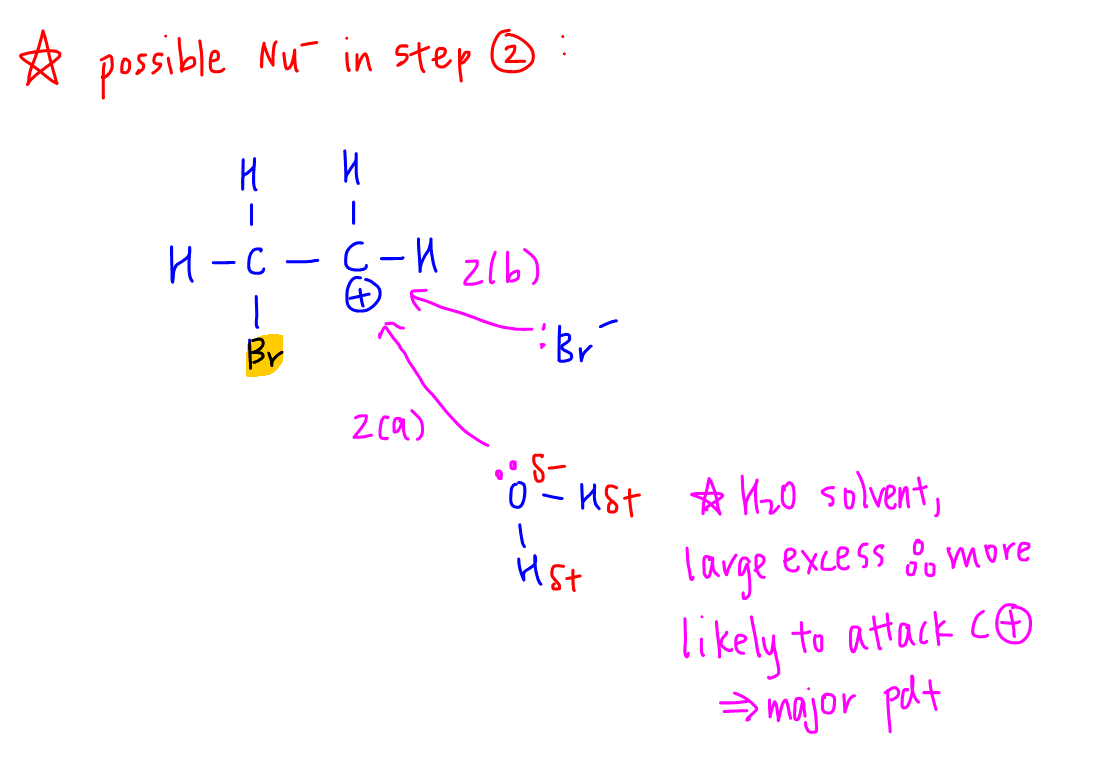
The probability of water attacking the carbocation is much higher since water is the solvent so there will be significantly more water as compared to Br-.
This explains why 2-bromoethanol is the major product and 1,2-dibromothane is the minor product.
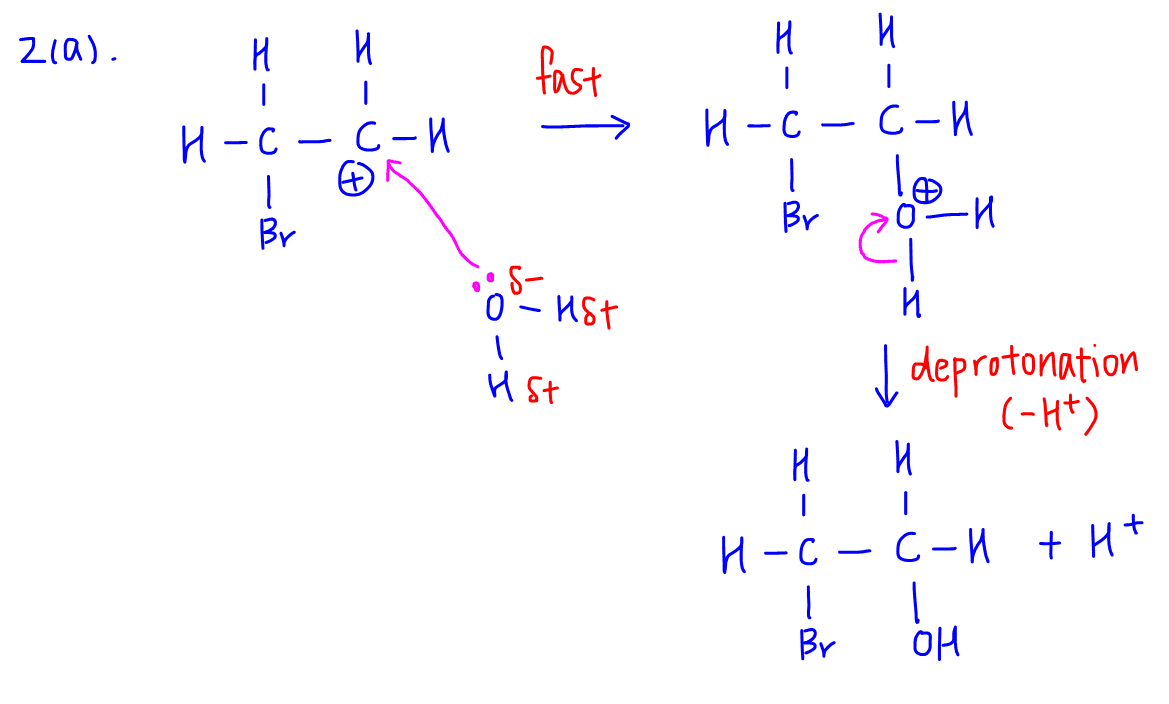
Formation of 2-bromoethanol involves water attacking the carbocation followed by deprotonation.
1,2-dibromoethane is formed when Br- attacks the carbocation.

Finally, an appreciation and understanding of electrophilic addition mechanism allows us to predict the possible products formed.
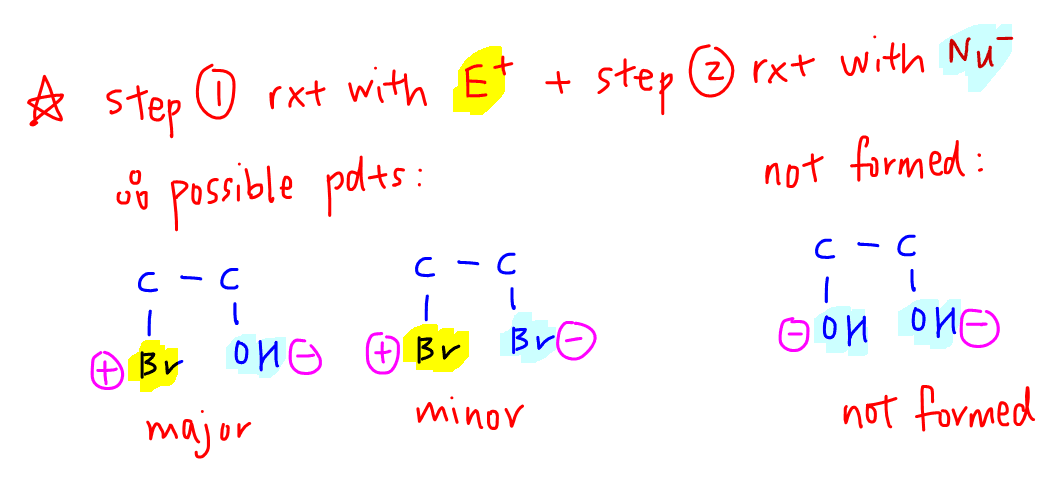
Ethan-1,2-diol is not formed since both oxygens are nucleophilic and cannot react with alkene as electrophiles.
Topic: Alkenes, Organic Chemistry, A Level Chemistry, Singapore
Back to other previous Organic Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's trusted JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
