Determine Enthalpy Change Using Average Bond Energy
Let Chemistry Guru, Singapore's top JC Chemistry tuition centre, guide you through an example where we have to determine the enthalpy change of the following reaction using average bond energy values from the Data Booklet:
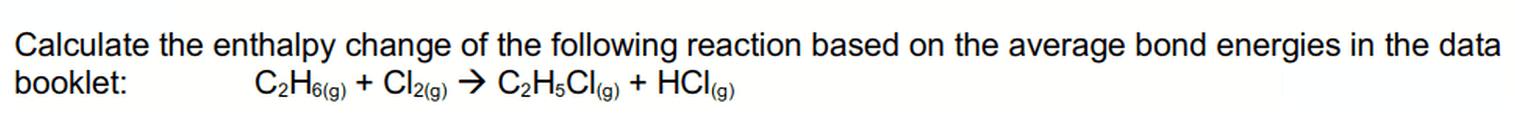
In a chemical reaction, bonds in the reactants are broken and bonds in the products are formed.
Therefore by comparing the bond energies of the bonds broken in the reactants and the bond energies of the bonds formed in the products, we can calculate the enthalpy change of the reaction.
First we draw the displayed formula for all the reactants and products so it will be easier to count the number of bonds involved.

Then we have to consider all the bonds broken in the reactants and work out the total energy involved by referring to the data booklet.

In this case the magnitude of all the bond energies required to break all the bonds in the reactants will be 3054 kJ mol-1.
Next we do the same for all the bonds formed in the products and determine the total energy involved.

The magnitude of all the bond energies released from forming all the bonds in the products will be 3171 kJ mol-1.
Finally we can use the follow formula to determine the enthalpy change of reaction.

Notice the formula will automatically convert the bond energies of bonds broken in reactants to an endothermic term (+ve) and the bond energies of bonds formed in products to an exothermic term (-ve).
Limitations of Average Bond Energy Calculations
This simple method to calculate enthalpy change of a reaction using average bond energies has the following limitations:
1. The bond energy values in the data booklet are average values hence calculations are estimates only.
2. The calculation is for gaseous species (reactants and products) only hence the calculated answer for reactions that involve solids and liquids will be even less accurate.

Topic: Energetics, Physical Chemistry, A Level Chemistry, Singapore
Back to other previous Physical Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's renowned A Level Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my A Level H2 Chemistry Tuition classes at Bishan or online tuition classes!
