2019 A Level H2 Chemistry Paper 1 Question 13 - Explain H2O2 Decomposition via Thermodynamics
Here's 2019 A Levels H2 Chemistry Paper 1 Question 13.

We need to explain why hydrogen peroxide can be stored for several weeks without much decomposition.
Let Chemistry Guru, Singapore's renowned JC Chemistry tuition centre, guide you through each statement and see if it can explain this observation.
3. The entropy change for the reaction at 20oC is negative.
We will consider this first since it is easier to deduce change in entropy.
By looking at the overall equation we see that there is an increase in the moles of gases formed.
This means there is an increase in the degree of disorder, hence entropy change is positive.
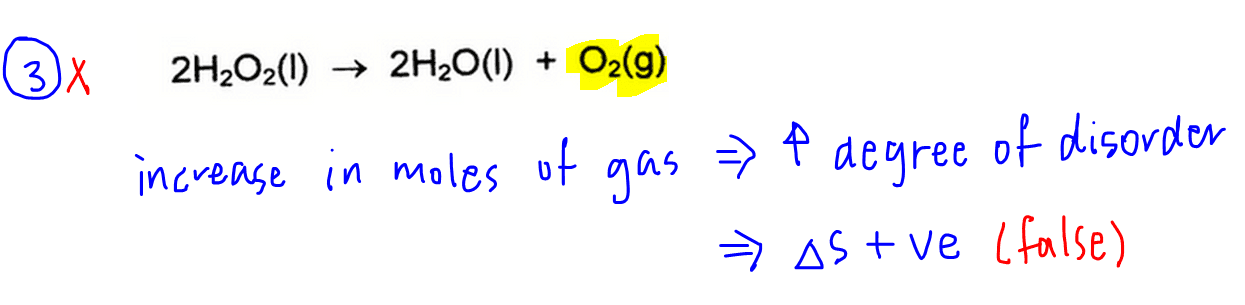
Therefore statement 3 is false.
1. The Gibbs free energy change for the reaction at 20oC is positive.
With the sign of entropy change deduced and sign of enthalpy change given, we can now deduce the sign of Gibbs free energy.
Since entropy change is positive, -TΔS is always negative.
Since ΔH is also negative, ΔG is always negative at all temperatures.

Hence statement 1 is false.
2. The reaction has a very high activation energy.
Activation energy is related to kinetics and rate of reaction.
There is no kinetics information in this question hence we cannot determine if statement 2 is true or not.
However if it were true, then a very high activation energy means the rate of H2O2 decomposition is very slow.
This would explain the observation that H2O2 can be stored for several weeks.
Since statements 1 and 3 are false, then in order for us to answer this question, statement 2 has to be true.

Therefore the answer to this question will be option D.
Topic: Energetics, Physical Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2019 A Level H2 Chemistry Paper 1
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's top H2 Chemistry tuition choice since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or online tuition classes!
