How to Determine pH of Weak Acid - Ionic Equilibria
In this video we want to learn how to determine the pH of a weak acid given its concentration and acid dissociation constant.
Let Chemistry Guru, Singapore's top JC Chemistry tuition centre, guide you through this exercise.
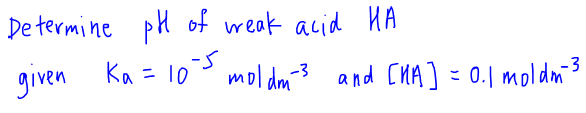
The first thing we can do is to write out the dissociation of the weak acid in solution and to complete the ICE table.

The ICE table is a summary to represent the changes in concentrations of reactants and products, where:
I - initial concentration
C - change in concentration
E - equilibrium concentration
So from the ICE table we know that at equilibrium,
[HA] is (0.1 - x) mol dm-3
[H+] is x mol dm-3
[A-] is x mol dm-3
We can then write out the acid dissociation constant term Ka and substitute in the concentrations to get a expression between Ka and x.
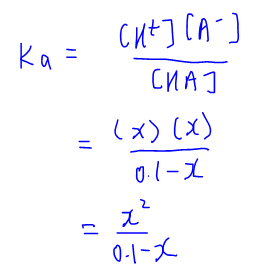
Notice that this is a quadratic expression and since for A Level H2 Chemistry we are not required to solve quadratic equations, we need to impose an approximation to simplify the Ka expression.
The approximation is very simple. Since HA is a weak acid with a small degree of dissociation, x is small therefore (0.1 - x) is approximately equal to 0.1.
We can then solve for x which is the concentration of H+ and is calculated to be 10-3 mol dm-3.
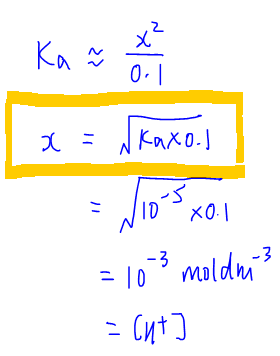
Finally we can calculate pH of this weak acid to be 3 using the following expression.
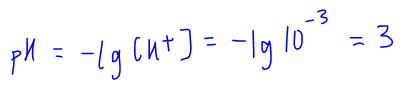
Usually we can apply the ICE table and approximation in a standardised way so we can derive this outcome to quickly determine H+ concentration given Ka and concentration of a weak acid:

Similarly we can have an expression to determine the OH- concentration of a weak base:
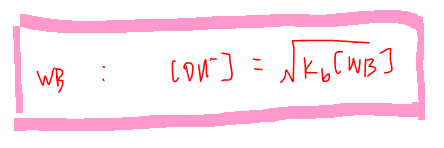
For the detailed step-by-step discussion on how to determine the weak acid's pH, check out this video!
Topic: Ionic Equilibria, Physical Chemistry, A Level Chemistry, Singapore
Back to other previous Physical Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's leading JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my A Level H2 Chemistry Tuition classes at Bishan or online chemistry classes!
