Limitation of Markovnikov Rule
In this JC1 webinar we want to understand Markovnikov Rule in depth by discussing its limitations.
Recall Markovnikov Rule states that the hydrogen will join to the carbon with more hydrogen to form major product.
If you are not familiar with this rule for electrophilic addition of alkenes, please view this video explaining Markovnikov Rule in detail.
Let's consider addition of HBr to the following alkene and try to deduce the major product.
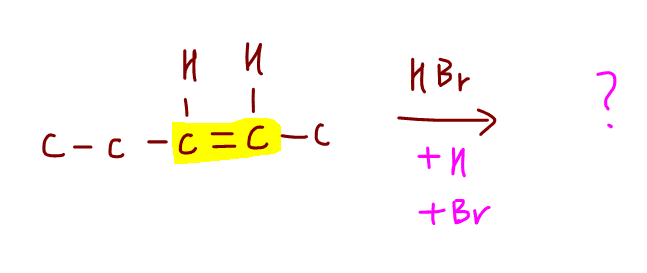
Notice for both alkene carbons, each carbon is bonded to 1 hydrogen and 1 alkyl group.
In this scenario, Markovnikov Rule is not useful to deduce the major product, as the rule requires the alkene carbons to have different number of hydrogen.
Can we then deduce the major product by comparing the stability of the carbocations formed during electrophilic addition?
Let's draw the first step of the EA mechanism to find out.
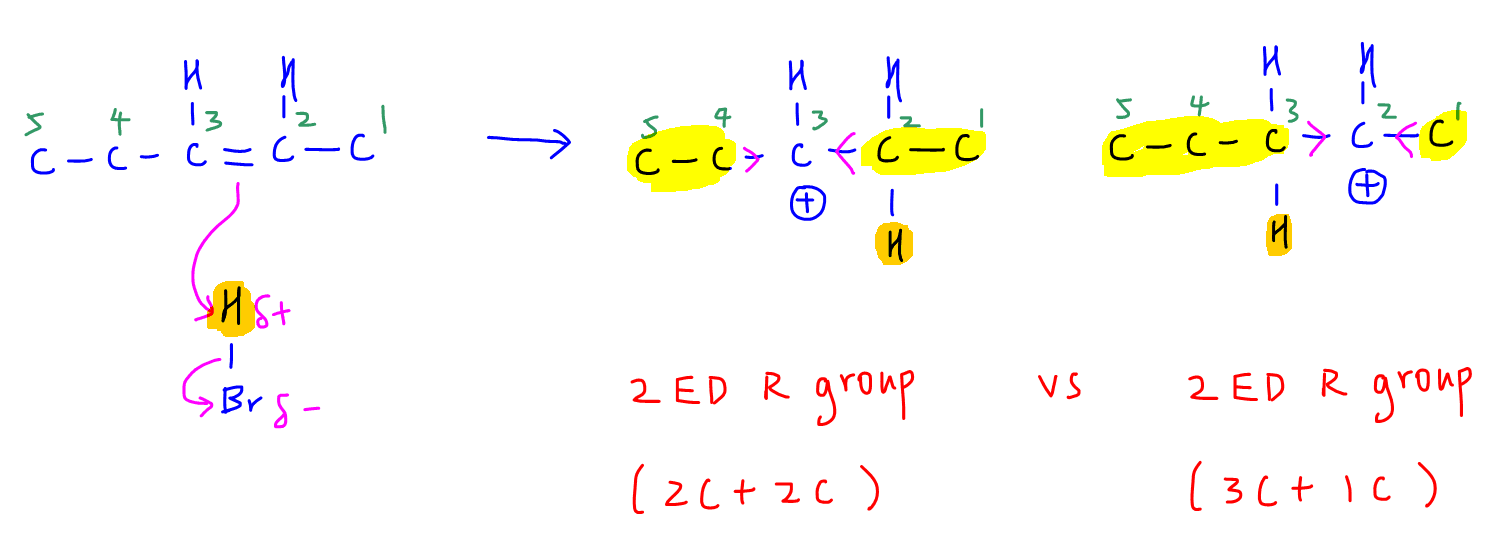
From the above step we have 2 different carbocations formed when H from HBr is added to the alkene.
The carbocation on the left is attached to 2 electron donating groups - both ethyl groups.
The carbocation of the right is also attached to 2 electron donating groups - 1 propyl group and 1 methyl group.
The bigger the alkyl group, the more electron donating it is.
The electron donating effect for the alkyl groups will increase in the order: methyl, ethyl, propyl.
Therefore we cannot compare the overall donating effect of 2 ethyl groups versus 1 propyl (more donating than ethyl) and 1 methyl (less donating than ethyl) group.
This is the reason why we are unable to determine which carbocation is more stable and give us the major product.
Topic: Alkenes, Organic Chemistry, A Level Chemistry, Singapore
Back to other previous Organic Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's trusted JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
