2019 A Level H2 Chemistry Paper 1 Question 29 - Measure Standard Electrode Potential
Let Chemistry Guru, Singapore's renowned JC Chemistry tuition centre, guide you through 2019 A Level H2 Chemistry Paper 1 Question 29.
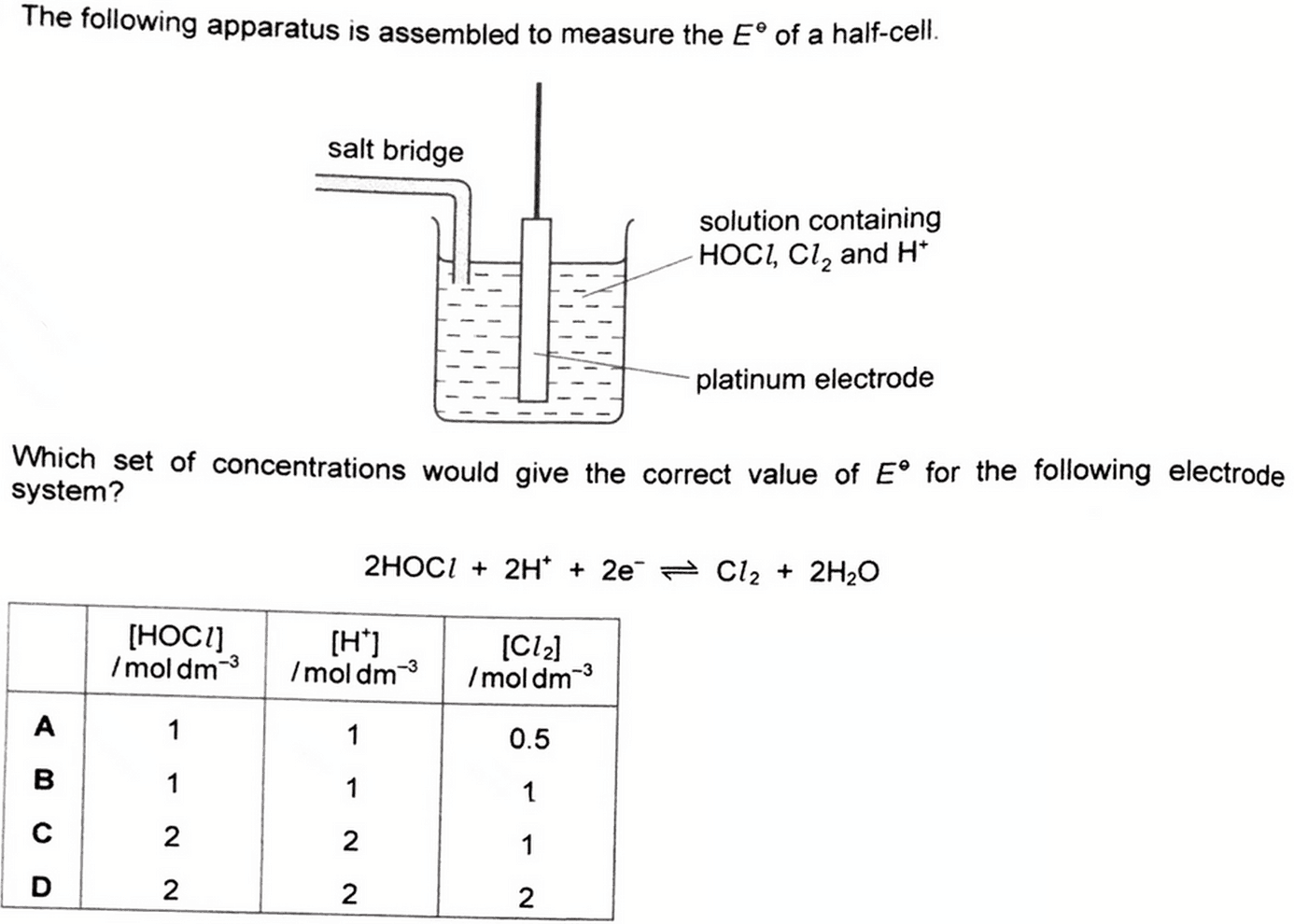
We are asked to determine the set of concentrations that would give the correct value of standard electrode potential for an electrode system.
First we will have to determine which species are taking part in reduction/oxidation in this reversible electrode.
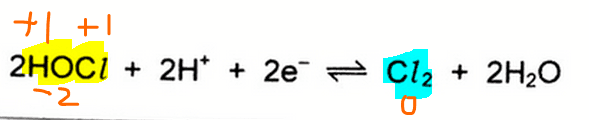
Notice the oxidation state of Cl is +1 in HOCl and zero in Cl2.
This means the species that we should focus on is HOCl and Cl2 since there is a change on oxidation state for Cl.
In the forward direction HOCl(+1) is reduced to Cl2(0), while in the reverse direction Cl2(0) is oxidised to HOCl(+1).
The standard conditions required for standard electrode potential should apply to both HOCl and Cl2.
Standard conditions: Temperature at 298K, Pressure at 1 atm, Concentration of solutions at 1.0 moldm-3
Hence concentration of HOCl and Cl2 has to be both at 1.0 moldm-3.
Notice the concentration of H+ is not that important since it just provides the acidic medium for the system.

By comparing the options we can conclude the correct answer to this question should be option B.
Topic: Electrochemistry, Physical Chemistry, A Level Chemistry, Singapore
Back to list of questions for 2019 A Level H2 Chemistry Paper 1
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's top H2 Chemistry tuition choice since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or online tuition classes!
