Can Nickel Spatula Stir Copper Sulfate Solution?
A nickel spatula is a common lab apparatus used to stir solutions.
Copper (II) sulfate solution is a blue solution that we can easily prepare in the lab.
So can we use a nickel spatula to stir a solution of copper (II) sulfate?

To answer this we have to consider the stability of nickel spatula when mixed with CuSO4 solution.
If nickel is stable in CuSO4, a nickel spatula can be used for stirring as it will not have any unintended reaction taking place.
If nickel is not stable in CuSO4, a reaction between nickel and CuSO4 will occur.
Then a nickel spatula will not be suitable for stirring.
Stability of nickel in CuSO4 is essentially the feasibility of the redox reaction between Ni and Cu2+.
So the real question is:
Is the redox reaction between Ni and Cu2+ feasible?
To determine this we can calculate the Ecell for this redox reaction.
Let's find the appropriate half equations in the data booklet:
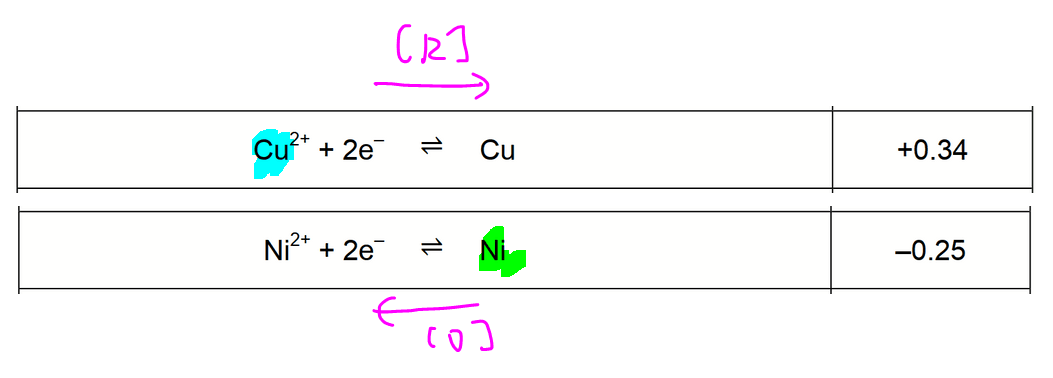
This redox reaction involves:
- reduction of Cu2+ to Cu E = +0.34V
- oxidation of Ni to Ni2+ E = -0.25V
We can now calculate the Ecell of this reaction:

Since Ecell is positive, the reaction between Ni and Cu2+ is feasible.
This means Ni is not stable in Cu2+ and we cannot use a nickel spatula to stir a solution of CuSO4.
Topic: Electrochemistry, Physical Chemistry, A Level Chemistry, Singapore
Back to other previous Physical Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's esteemed H2 Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
