Saytzeff Rule, Zaitsev Rule: How to predict Major Product for Elimination Reaction
Elimination Reaction for some alcohols and alkyl halides will result in different alkene products, and Saytzeff or Zaitsev Rule is used to determine the major product.
Saytzeff or Zaitsev Rule states that the more substituted alkene will be the major product.
So by looking at the number of alkyl groups attached to the alkene, the degree of substitution and hence major and minor products can be determined.
Let's take the elimination of 2-bromobutane as an example:
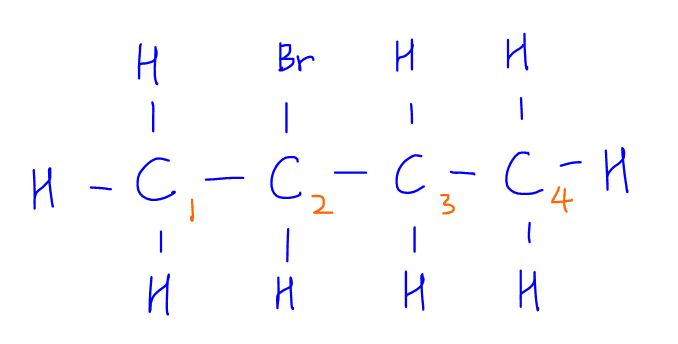
We notice that when bromine is eliminated from carbon-2, we can either remove a neighbouring hydrogen from carbon-1 or carbon-3.
Therefore 2 different alkene products, but-1-ene and but-2-ene can be formed.
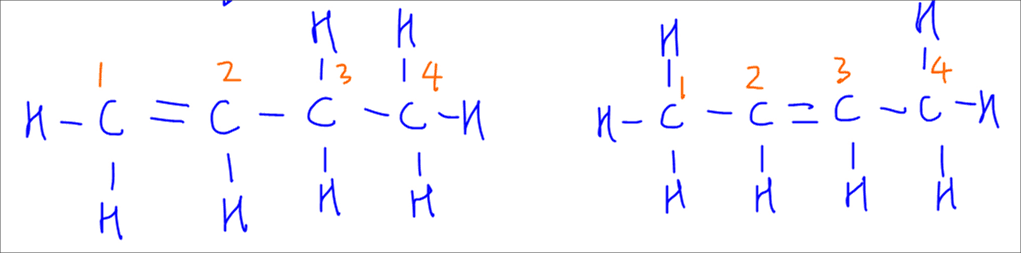
To determine which is the major product, we have to figure out the degree of substitution for each alkene.
To make it easier to visualise, we can box up the alkene and count the number of alkyl groups attached to the alkene.
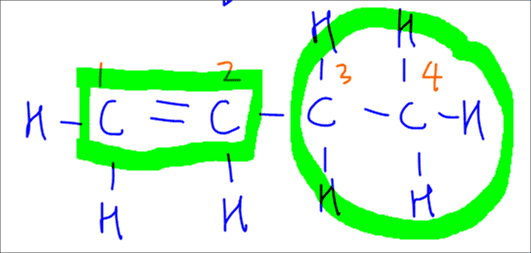
For but-1-ene we notice it has only 1 alkyl group attached to the alkene. It is less substituted hence the minor product.
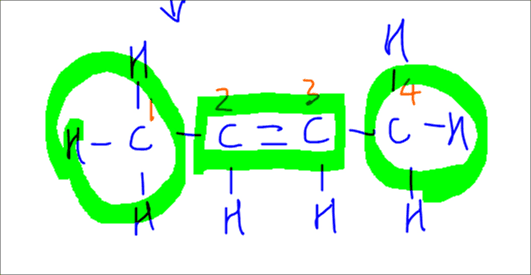
For but-2-ene, it has 2 alkyl groups attached to the alkene. It is more substituted therefore the major product.
For a step-by-step guide to predict the major product using Saytzeff Rule, check out this video lesson now!
Topic: Alkenes, Organic Chemistry, A Level Chemistry, Singapore
Back to other previous Organic Chemistry Video Lessons.
A Level H2 Chemistry Video Lessons
Chemistry Guru, Singapore's reputable JC Chemistry tuition centre, has a huge collection of short video lessons that targets important H2 Chemistry concepts and common questions.
Join my 19,000 subscribers on my YouTube Channel for new video lessons every week!
Follow me on Instagram for H2 Chemistry videos and (not so funny) memes!
You can also view other A Level H2 Chemistry videos here at my website.
Need an experienced tutor to make Chemistry simpler for you?
My weekly classes in Singapore are ideal for students who prefer a more structured program.
Build a strong foundation and ace your exams!
Sign up now for a trial lesson at $50only!
Check out our Chemistry tuition class timing and topics covered for our popular JC1 Classes and JC2 Classes at Bishan, Singapore.
Online lessons are also available! Learn H2 Chemistry anytime, anywhere at 50% of the cost of conventional class tuition.
Find out more information about our online tuition.
