Strong Oxidation of Alkene and Alkylbenzene
Let Chemistry Guru, Singapore's esteemed A Level Chemistry tuition centre, guide you through our discussion question this week!
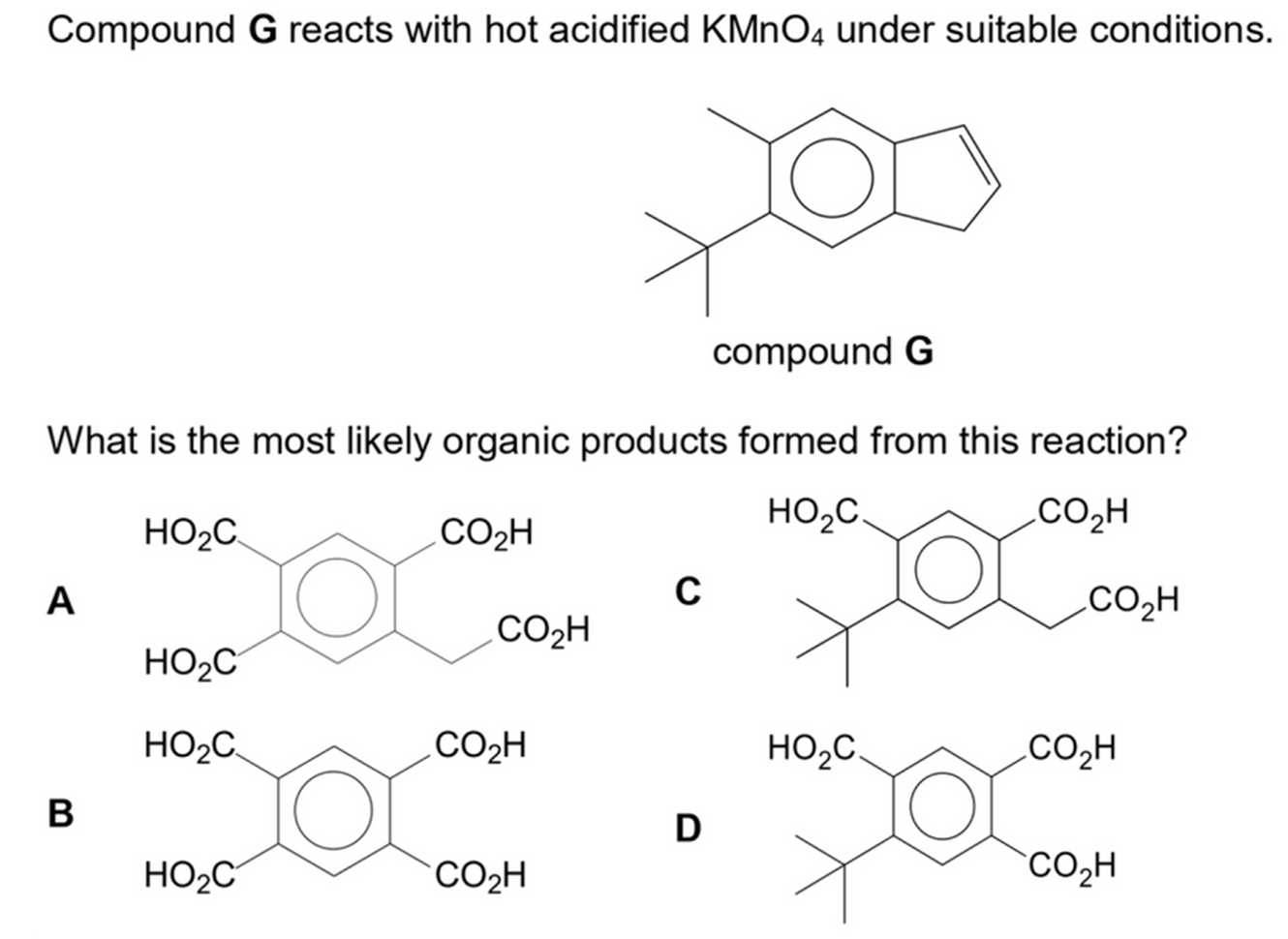
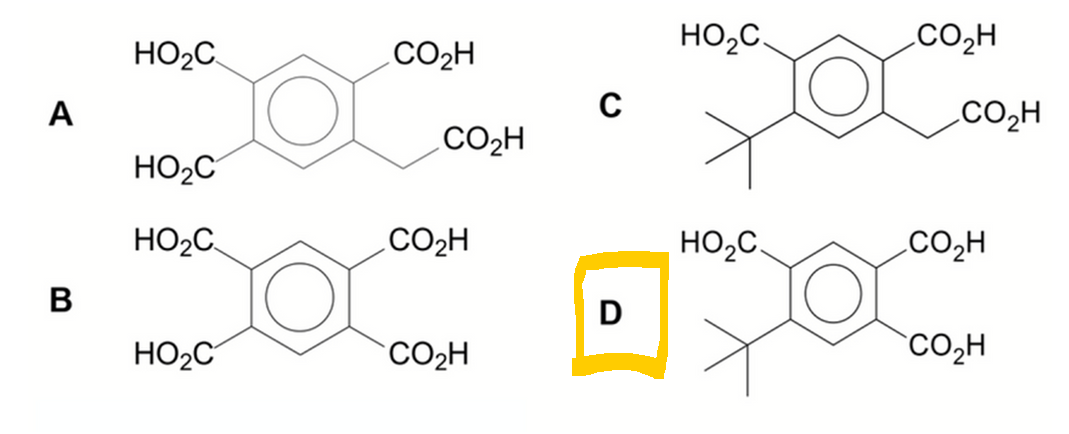
We want to determine the product formed when compound G is oxidised with hot acidified KMnO4.
Functional groups present are alkene and alkylbenzene.
Let's recap their oxidation first.
1. Strong oxidation of alkenes
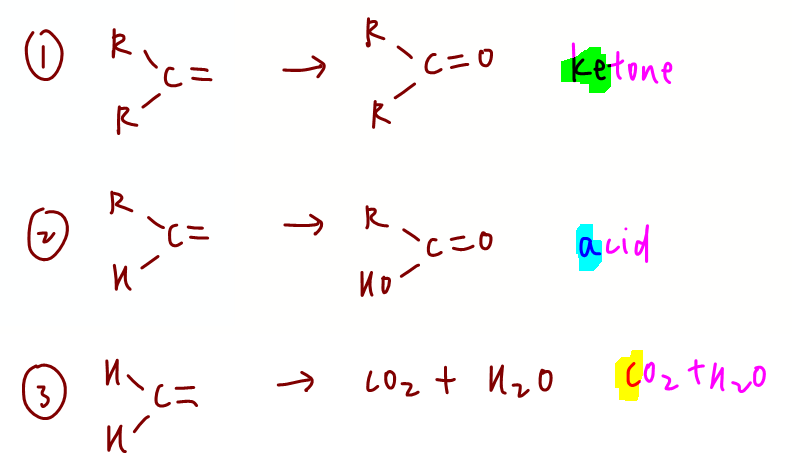
We have recap this in our video lesson last week.
2. Strong oxidation of alkylbenzene
If carbon directly attached to benzene has minimum 1 hydrogen, it will be oxidised to benzoic acid.
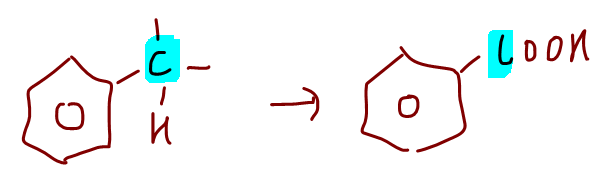
Check out this video lesson for in-depth discussion of strong oxidation of alkylbenzene.
Let Chemistry Guru, Singapore's esteemed A Level Chemistry tuition centre, guide you through compound G and deduce the product.
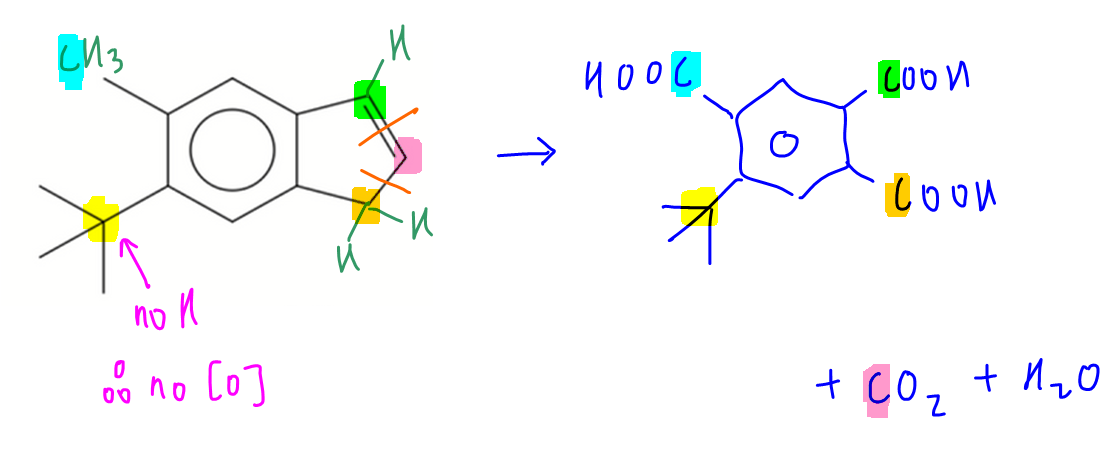
Blue carbon is a methyl group with 3 hydrogens so it will be oxidised to acid group.
Yellow carbon is a quaternary carbon hence it cannot be oxidised.
Green carbon is alkene carbon attached to benzene and hydrogen so will be oxidised to acid group.
Orange carbon is alkyl carbon with 2 hydrogens so it will be oxidised to acid group.
This will result in the breaking of C-C bond between orange and pink carbons.
Finally the pink carbon will be isolated due to oxidation of alkene and oxidation of orange alkyl carbon.
This pink carbon will be oxidised to carbon dioxide and water.
Hence the answer to this question will be option D.
Topic: Benzene, Organic Chemistry, A Level Chemistry, Singapore
Back to other previous Organic Chemistry Video Lessons.
Found this A Level Chemistry video useful?
This free chemistry video lesson is brought to you by Chemistry Guru, Singapore's top JC Chemistry tuition centre since 2010.
Please like this video and share it with your friends!
Join my 19,000 subscribers on my YouTube Channel for new A Level Chemistry video lessons every week.
Check out other A Level Chemistry Video Lessons here!
Need an experienced tutor to make Chemistry simpler for you?
Do consider signing up for my JC Chemistry Tuition classes at Bishan or on-demand video lessons!
